Utilization of Aluminon for Determination of Ultra-trace Levels of Copper in Environmental, Biological, and Water Samples via Cloud Point Extraction Coupled with Spectrophotometry

International Journal of Analytical and Bioanalytical Methods
(ISSN: 2633-8912)
Volume 2, Issue 1
Research Article
DOI: 10.35840/2633-8912/2413
Utilization of Aluminon for Determination of Ultra-trace Levels of Copper in Environmental, Biological, and Water Samples via Cloud Point Extraction Coupled with Spectrophotometry
Ragab Y Sharaf, Waheed M Salem, Alaa E Ali and Alaa S Amin
Table of Content
Figures
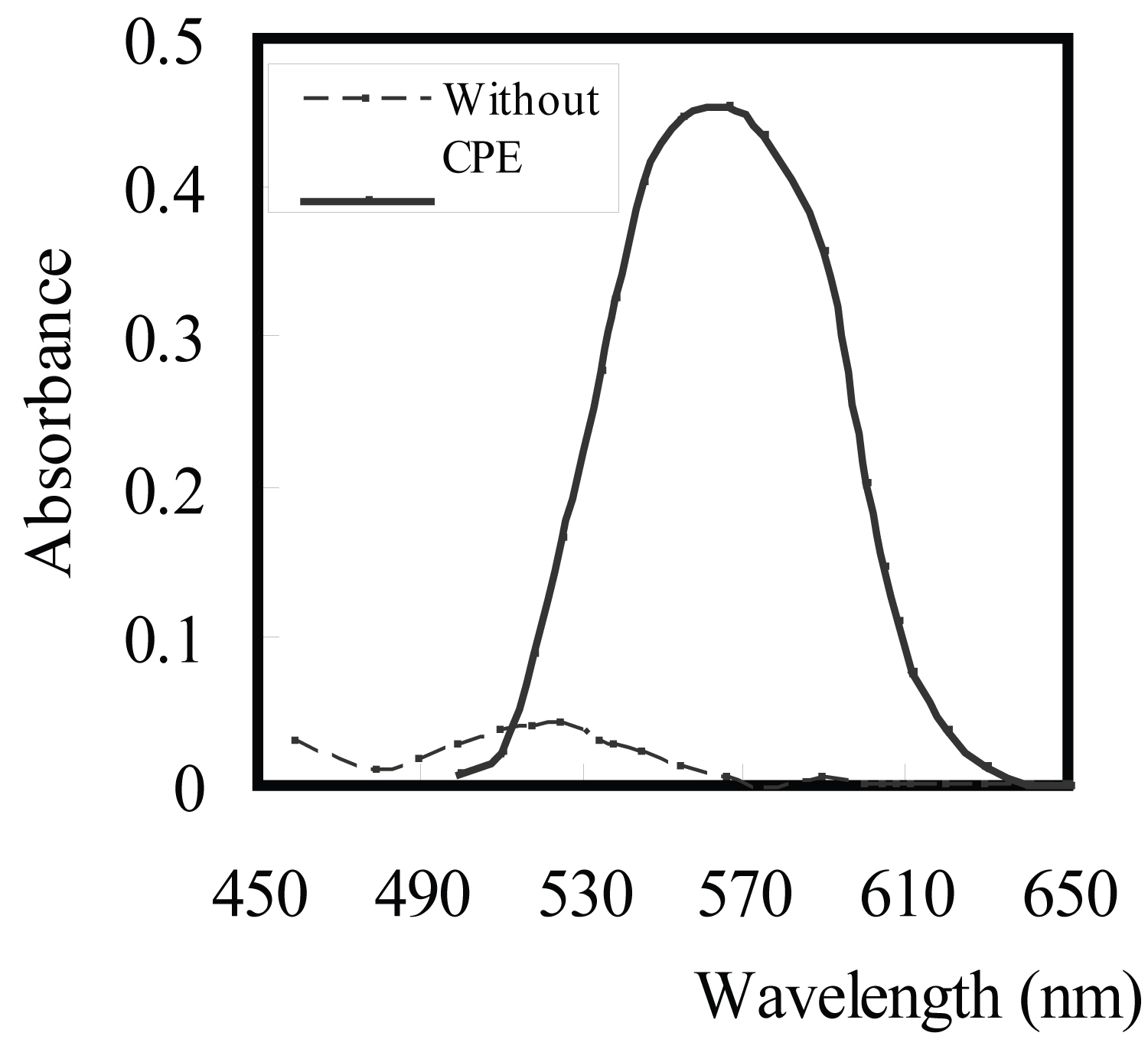
Figure 1: Absorption spectra for 150 ng/ml Cu2+ complexed....
Absorption spectra for 150 ng/ml Cu2+ complexed with 2 × 10-5 M aluminon using 0.15% Triton X-114 at pH 3.5 with CPE and for 1.5 mg/ml without CPE.
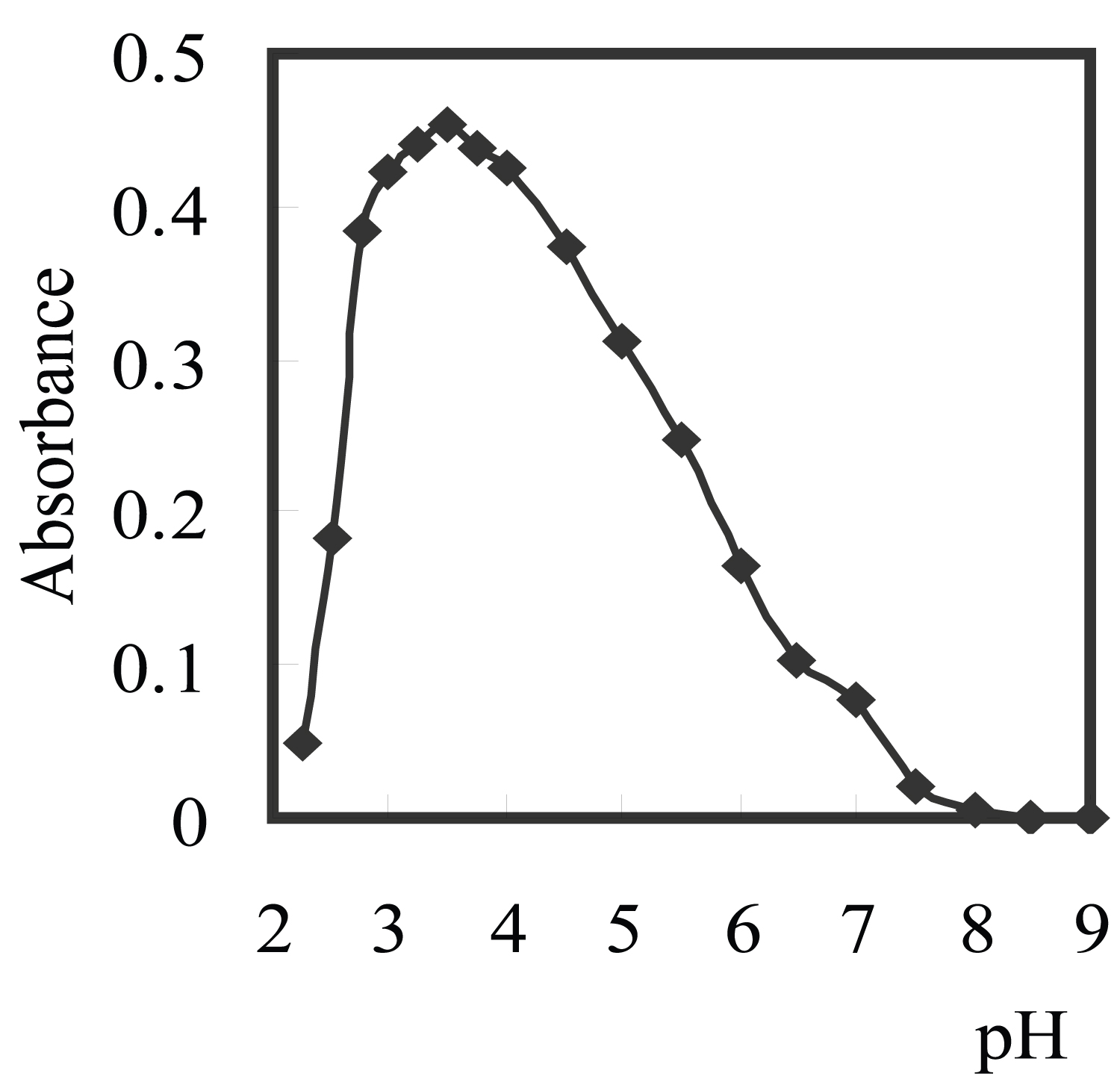
Figure 2: Effect of pH of on complexation of 150 ng/ml.....
Effect of pH of on complexation of 150 ng/ml Cu2+ with 1.5 × 10-5 M aluminon using 0.15% Triton X-114 (SD = ± 0.06).
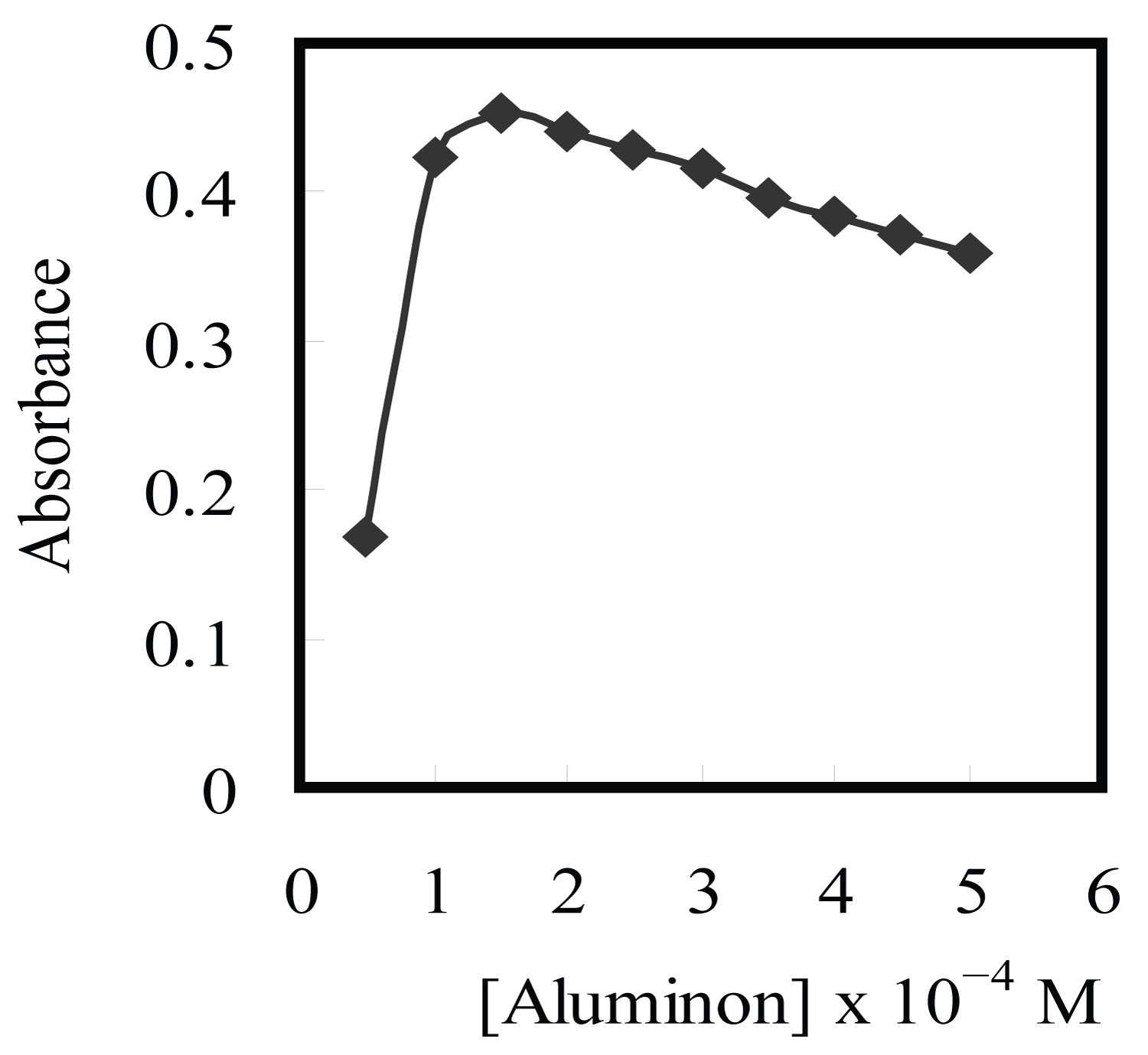
Figure 3: Effect of aluminon concentration on the....
Effect of aluminon concentration on the complexation of 150 ng/ml Cu2+ using 0.15% Triton X-114 at pH 3.5 (SD = ± 0.04).
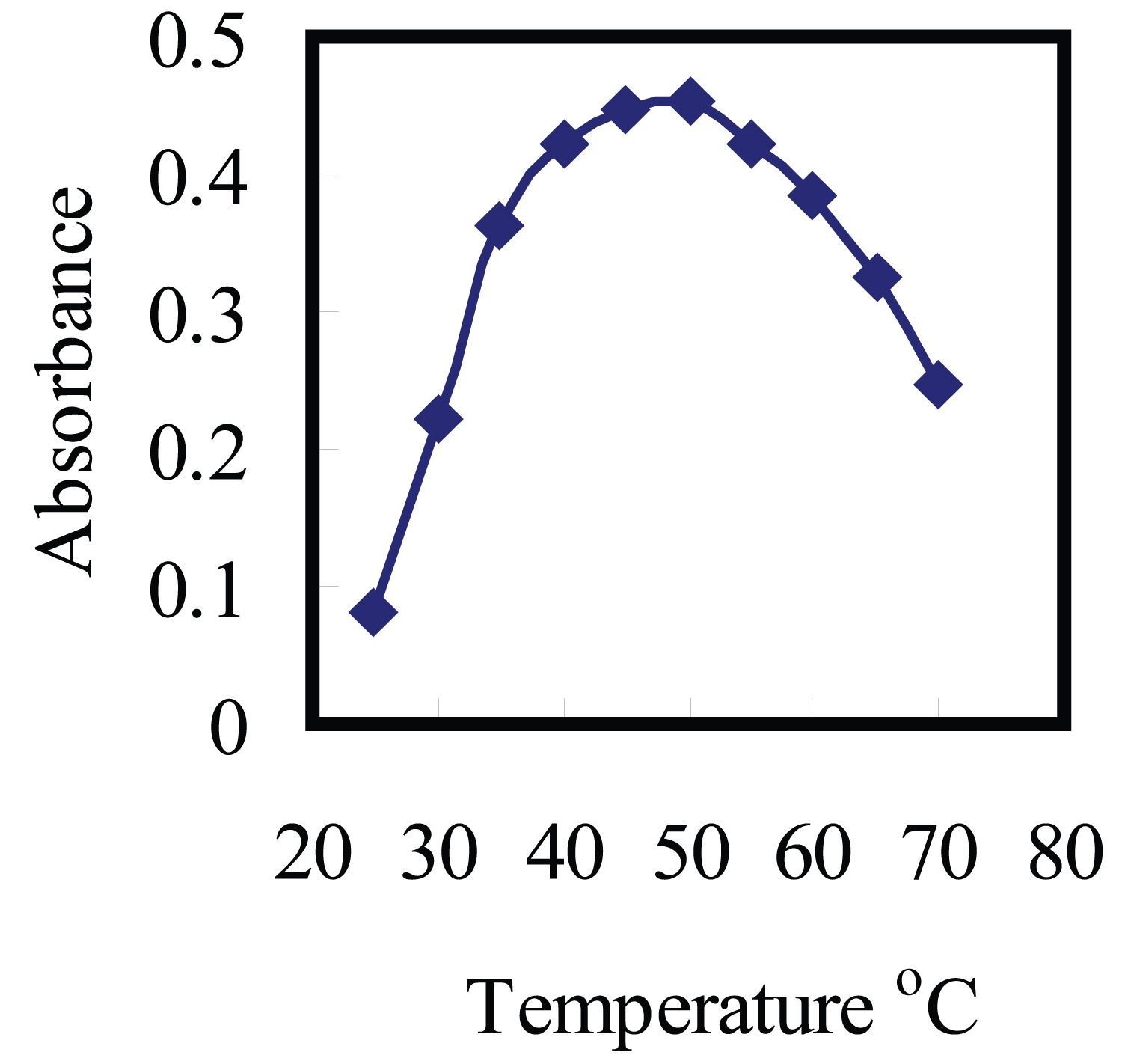
Figure 4: Temperature effect on the complexation of....
Temperature effect on the complexation of 150 ng/ml Cu(II) with 1.5 × 10-5 M aluminon using 0.15% Triton X-114.
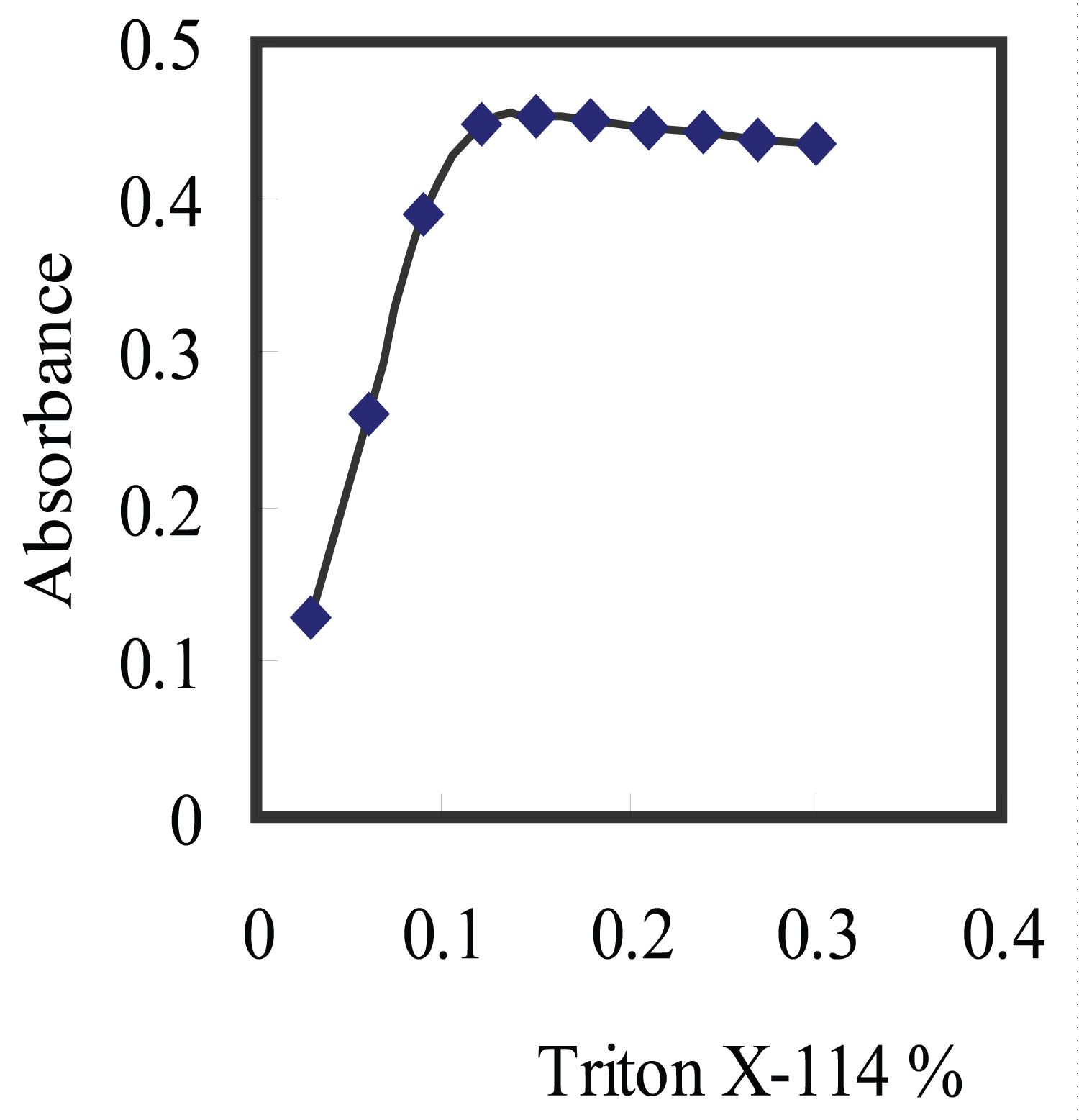
Figure 5: Effect of Triton X-114 concentration on the complexation....
Effect of Triton X-114 concentration on the complexation of 150 ng/ml Cu2+ with 2.0 × 10-4 M aluminon at pH 3.5 (SD = ± 0.04).
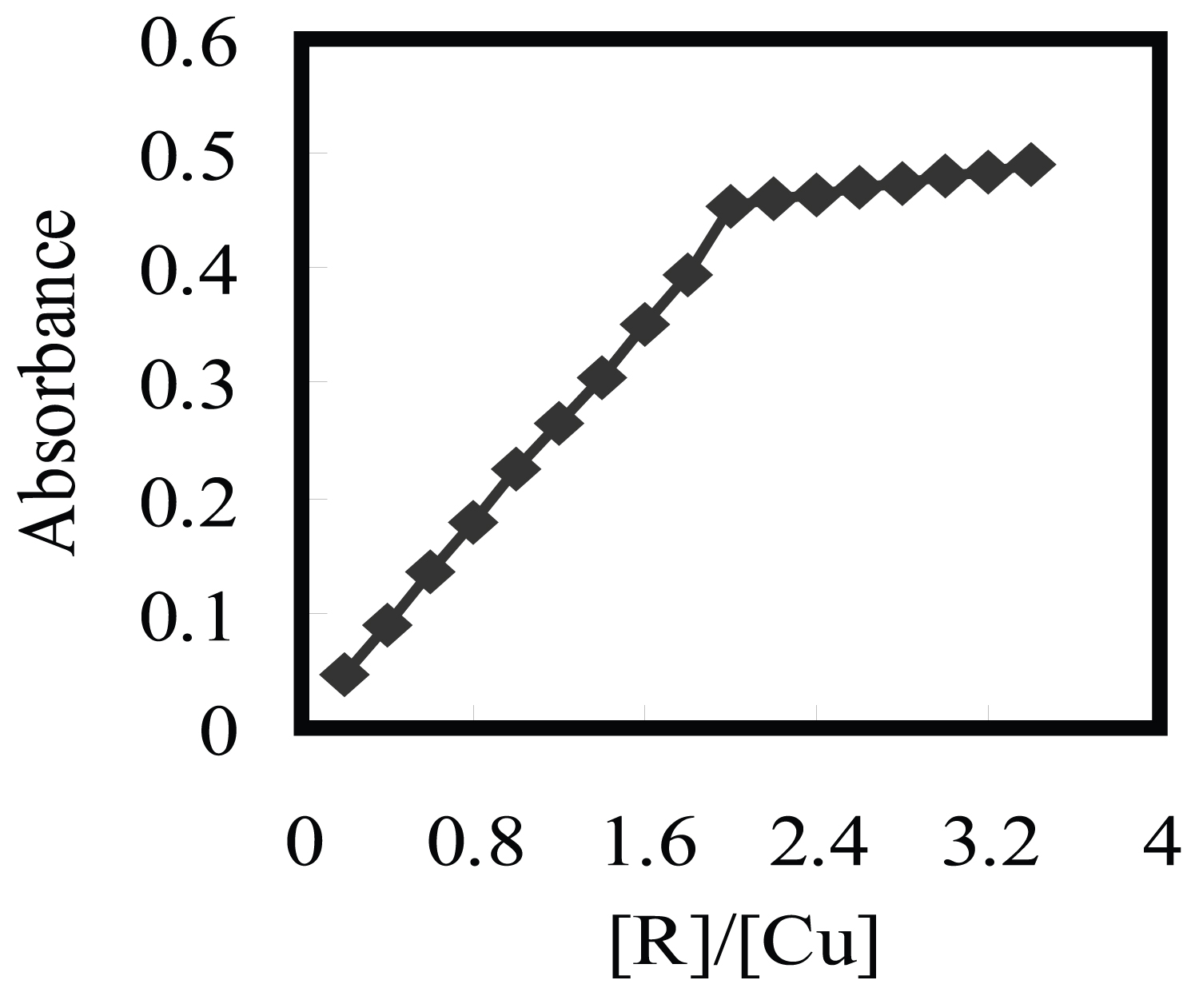
Figure 6: Molar ratio method for complexation of 2.0....
Molar ratio method for complexation of 2.0 × 10-4 M aluminon with Cu2+ at pH 3.5.
Tables
Table 1: Analytical features of the proposed method.
Table 2: Comparative data from some recent studies on preconcentration-separation of copper.
Table 3: Comparison of selected reagents for the spectrophotometric determination of copper.
Table 4: Tolerance ratio of diverse ions on the determination of 150 ng/ml Cu2+ (relative error ± 5.0 %).
Table 5: Recovery studies of copper in real samples.
References
- R Ansari, TG Kazi, MK Jamali, MB Arain, ST Sherazi, et al. (2008) Improved extraction method for the determination of iron, copper, and nickel in new varieties of sunflower oil by atomic absorption spectroscopy. J AOAC Int 91: 400-407.
- H Xu, LP Zeng, SJ Xing, YZ Xian, LT Jin (2008) Nafion-coated bismuth film electrodes for the determination of trace lead and cadmium in herbal medicines by anodic stripping voltammetry. Chin J Chem 26: 847-852.
- B Selvi, D Mendil (2009) Determination of heavy metal levels in aquatic plants collected from Hoyran lake in Isparta, Turkey. Asian J Chem 21: 2807-2812.
- E Kendüzler, AR Türker (2003) Atomic absorption spectrophotometric determination of trace copper in waters, aluminium foil and tea samples after preconcentration with 1-nitroso-2-naphthol-3,6-disulfonic acid on Ambersorb 572. Anal Chim Acta 480: 259-266.
- Tabrizi AB, Hazard J, Mater B (2007) Development of a cloud point extraction-spectrofluorimetric method for trace copper(II) determination in water samples and parenteral solutions. Journal of Hazardous Materials 139: 260-264.
- PD Ding, LM Niu, HQ Luo, NB Li (2007) Application of meso-2,3-dimercaptosuccinic acid self-assembled gold electrode for voltammetric determination of copper. Croat Chem Acta 80: 61-66.
- RA Nalawade, AM Nalawade, GS Kamble, MA Anuse (2015) Rapid, synergistic extractive spectrophotometric determination of copper(II) by using sensitive chromogenic reagent N?,N?-bis[(E)-5(4-fluorophenyl) methylidene] thiocarbonohydrazide. Spectrochim Acta A 146: 297-306.
- MD Frahani, F Shemirani, NF Ramandi, M Gharehbaghi (2015) Ionic liquid as a ferro fluid carrier for dispersive solid phase extraction of copper from food samples. Food Anal Methods 8: 1979-1989.
- S Prasad, T Halafihi (2003) Development and validation of catalytic kinetic spectrophotometric method for determination of Cu(II). Microchim Acta 142: 237-244.
- R Gürkan, N Altunay (2013) A reliable method of quantification of trace copper in beverages with and without alcohol by spectrophotometry after cloud point extraction. Quim Nova 36: 1146-1154.
- M Ghaedi, MR Fathi, F Marahel, F Ahmadi (2005) Development of efficient method for preconcentration and determination of copper, nickel, zinc and iron ions in environmental samples by combination of cloud point extraction and flame atomic absorption spectrometry. Central European Journal of Chemistry 7: 148-154.
- M Ghaedi, E Asadpour, A Vafaie (2006) Simultaneous preconcentration and determination of copper, nickel, cobalt, lead, and iron content using a surfactant-coated alumina. Bull Chem Soc Japan 79: 432-436.
- M Ghaedi, A Shokrollahi (2006) Copper concentration using micelle-mediated extraction in real samples. Fresen Environ Bull 15: 1373-1378.
- SA Reddy, KJ Reddy, SL Narayan, AV Reddy (2008) Analytical applications of 2, 6-diacetylpyridine bis-4-phenyl-3-thiosemicarbazone and determination of Cu(II) in food samples. Food Chem 109: 654-659.
- Camp Dresser, McKee, Inc (2004) Guidelines for water reuse. U.S. Environmental Protection Agency, EPA/625/R-04/108, (NTIS PB2005 106542), Washington, USA.
- Ministry of Science, Technology and Environment (2006) Thailand State of Pollution Report. Bangkok, Thailand.
- F Aguilar, UR Charrondiere, B Dusemund, P Galtier, J Gilbert, et al. (2009) Copper(II) oxide as a source of copper added for nutritional purposes to food supplements, scientific opinion of the panel on food additives and nutrient sources added to food. EFSA J 1089: 1-15.
- Y Orooji, M Ghanbari, O Amiri, M Salavati-Niasar (2020) Facile fabrication of silver iodide/graphitic carbon nitride nanocomposites by notable photo-catalytic performance through sunlight and antimicrobial activity. J Hazard Mat 389: 122079.
- M Ghanbari, M Salavati-Niasar (2018) Tl4CdI6 nanostructures: Facile sonochemical synthesis and photocatalytic activity for removal of organic dyes. Inorg Chem 57: 11443-11455.
- M Karami, M Ghanbari, O Amiri, M Salavati-Niasar (2020) Enhanced antibacterial activity and photocatalytic degradation of organicdyes under visible light using cesium lead iodide perovskite nanostructures prepared by hydrothermal method. Sep & Pur Tech 253: 117526.
- S Gholamrezaei, M Amiri, O Amiri, M Salavati-Niasari, H Moayedi (2020) Ultrasound-accelerated synthesis of uniform SrMnO3 nano- particles as water-oxidizing catalysts for water splitting systems. Ultrason Sonochem 62: 104899.
- XX Tian, S Gholamrezaei, O Amiri, M Ghanbari, A Dashtbozorg, et al. (2020) Zn2MnO4/ZnO nanocomposites: One step sonochemical fabrication and demonstration as a novel catalyst in water splitting reaction. Ceramics Int.
- M Behbahani, M Salarian, MM Amini, O Sadeghi, A Bagheri, et al. (2013) Application of a new functionalized nanoporous silica for simultaneous trace separation and determination of Cd(II), Cu(II), Ni(II) and Pb(II) in food and agricultural products. Food Anal Methods 6: 1320-1329.
- H Ebrahimzadeh, M Behbahani, Y Yamini, L Adlnasab, AA Asgharinezhad (2013) Optimization of Cu(II)-ion imprinted nanoparticles for trace monitoring of copper in water and fish samples using a Box-Behnken design. React Funct Polym 73: 23-29.
- Y Liu, P Liang, L Guo (2005) Nanometer titanium dioxide immobilized on silica gel as sorbent for preconcentration of metal ions prior to their determination by inductively coupled plasma atomic emission spectrometry. Talanta 68: 25-30.
- FH Quina, WL Hinze (1999) Surfactant-mediated cloud point extractions: an environmentally benign alternative separation approach. Ind Eng Chem Res 38: 4150-4168.
- MA Bezerra, RE Bruns, SLC Ferreira (2006) Statistical design-principal component analysis optimization of a multiple response procedure using cloud point extraction and simultaneous determination of metals by ICP OES. Anal Chim Acta 580: 251-257.
- H Filik, F Dondurmacioglu, R Apak (2008) Micelle mediated extraction of cadmium from water and tobacco samples with glyoxal-bis (2-hydroxyanil) and determination by electrothermal atomic absorption spectrometry. Int J Environ Anal Chem 88: 637-648.
- YJ Li, B Hu, ZC Jiang (2006) On-line cloud point extraction combined with electrothermal vaporization inductively coupled plasma atomic emission spectrometry for the speciation of inorganic antimony in environmental and biological samples. Anal Chim Acta 576: 207-214.
- M Ghaedi, A Shokrollah, R Mehrnoosh, O Hossaini, M Soylak (2008) Combination of cloud point extraction and flame atomic absorption spectrometry for preconcentration and determination of trace iron in environmental and biological samples. Cent Eur J Chem 6: 488-496.
- DLG Borges, MAMS da Veiga, VLA Frescura, B Welz, AJ Curtius (2003) Cloud-point extraction for the determination of Cd, Pb and Pd in blood by electrothermal atomic absorption spectrometry, using Ir or Ru as permanent modifiers. J Anal At Spectrom 18: 501-507.
- TA Maranhão, DLG Borges, MAMS Veiga, AJ Curtius (2005) Cloud point extraction for the determination of cadmium and lead in biological samples by graphite furnace atomic absorption spectrometry. Spectrochim Acta B 60: 667-672.
- F Shemirani, SD Abkenar, AA Mirroshandel, MS Niasari, RR Kozania (2003) Preconcentration and speciation of chromium in water samples by atomic absorption spectrometry after cloud-point extraction. Anal Sci 19: 1453-1456.
- H Sang, P Liang, D Du (2008) Determination of trace aluminum in biological and water samples by cloud point extraction preconcentration and graphite furnace atomic absorption spectrometry detection. J Hazard Mater 154: 1127-1132.
- K Kiran, KS Kumar, B Prasad, K Suvardhan, LR Babu, et al. (2008) Speciation determination of chromium (III) and (VI) using preconcentration cloud point extraction with flame atomic absorption spectrometry (FAAS). J Hazard Mater 150: 582-586.
- JL Manzoori, H Abdolmohammad-Zadeh, M Amjadi (2007) Ultratrace determination of cadmium by cold vapor atomic absorption spectrometry after preconcentration with a simplified cloud point extraction methodology. J Hazard Mater 144: 458-463.?
- MA Mesquita da Silva, VLA Frescura, AJ Curtius (2001) Determination of noble metals in biological samples by electrothermal vaporization inductively coupled plasma mass spectrometry, following cloud point extraction. Spectrochim Acta (B) 56: 1941-1949.
- L Tavakoli, Y Yamini, H Ebrahimzadeh, A Nezhadali, S Shariati (2008) Development of cloud point extraction for simultaneous extraction and determination of gold and palladium using ICP-OES. J Hazard Mater 152: 737-743.
- N Shokoufi, F Shemirani (2007) Laser induced-thermal lens spectrometry after cloud point extraction for the determination of trace amounts of rhodium. Talanta 73: 662-667.
- A Safavi, H Abdollahi, MR Hormozi Nezhad, R Kamali (2004) Cloud point extraction, preconcentration and simultaneous spectrophotometric determination of nickel and cobalt in water samples. Spectrochim Acta (A) 60: 2897-2901.
- M Ghaedi, F Ahmadi, M Soylak (2007) Preconcentration and separation of nickel, copper and cobalt using solid phase extraction and their determination in some real samples. J Hazard Mater 147: 226-231.
- HTS Britton (1952) Hydrogen ions. (4th edn), Chapman and Hall, London.
- M Ghaedi, F Ahmadi, MR Fathi, F Marahel (2005) Simultaneous preconcentration and determination of copper, nickel, cobalt and lead ions content by flame atomic absorption spectrometry. Fresenius Environ Bull 14: 1158-1163.
- M Ghaedi, A Shokrollahi, AH Kianfar, AS Mirsadeghi, A Pourfarokhi, et al. (2008) The determination of some heavy metals in food samples by flame atomic absorption spectrometry after their separation-preconcentration on bis salicylaldehyde, 1,3 propandiimine (BSPDI) loaded on activated carbon. J Hazard Mater 154: 128-134.?
- A Shokrollahi, M Ghaedi, S Gharaghani, MR Fathi, M Soylak (2008) Cloud point extraction for the determination of copper in environmental samples by flame atomic absorption spectrometry. Quim Nova 31: 70-74.
- AR Rod, S Borhani, F Shemirani (2006) Cloud point preconcentration and flame atomic absorption spectrometry: Application to the determination of manganese in milk and water samples. Eur Food Res Technol 223: 649-653.
- JH Clint (1992) Surfactant aggregation. Blackie academic, Glasgow, UK, 154.
- PC Hiemenz, RH Rajagopalan (1997) Principles of colloid and surface chemistry. (3rd edn), Marcel Dekker Inc., New York, 377.
- IUPAC (1978) Nomenclature, Symbols, units and their usage in spectrochemical analysis II. Spectrochim Acta (B) 33: 241-245.
- G Kaya, M Yaman (2008) Online preconcentration for the determination of lead, cadmium and copper by slotted tube atom trap (STAT)-flame atomic absorption spectrometry. Talanta 75: 1127-1133.
- Y Yamini, M Faraji, S Shariati, R Hassani, M Ghanbarian (2008) On-line metals preconcentration and simultaneous determination using cloud point extraction and inductively coupled plasma optical emission spectrometry in water samples. Anal Chim Acta 612: 144-151.
- VA Lemos, MA Bezerra, FA Amorim (2008) On-line preconcentration using a resin functionalized with 3,4-dihydroxybenzoic acid for the determination of trace elements in biological samples by thermospray flame furnace atomic absorption spectrometry. J Hazard Mater 157: 613-619.
- VA Lemos, Gd Novaes, AL de Carvalho, EM Gama, AG Santos (2009) Determination of copper in biological samples by flame atomic absorption spectrometry after precipitation with Me-BTAP. Environmental Monitoring and Assessment 148: 245-253.
- CZ Huang, B Hu (2008) Silica-coated magnetic nanoparticles modified with ?-mercaptopropyltrimethoxysilane for fast and selective solid phase extraction of trace amounts of Cd, Cu, Hg, and Pb in environmental and biological samples prior to their determination by inductively coupled plasma mass spectrometry. Spectrochim Acta (B) 63: 437-444.
- A Shokrollahi, M Ghaedi, O Hossainia, N Khanjari, M Soylak (2008) Cloud point extraction and flame atomic absorption spectrometry combination for copper(II) ion in environmental and biological samples. J Hazard Mater 160: 435-440.
- VA Lemos, MS Santos, GT David, MV Maciel, MA Bezerra (2008) Development of a cloud point extraction method for copper and nickel determination in food samples. J Hazard Mater 159: 245-251.
- S Karabocek, S Nohut, O Dalman, S Guner (2000) A new spectrophotometric reagent for copper: 3,3'-(1,3-propanediyldi-imine)bis-[3-methyl-2-butanone]dioxime. Anal Chim Acta 408: 163-168.
- N Chimpalee, D Chimpalee, S Lohwithee, L Nakwatchara, DT Burns (1996) Spectrophotometric determination of copper after extraction of its chelate with bis(acetylacetone)ethylenedimine. Anal Chim Acta 329: 315-318.
- S Yi (1987) A study of the spectrophotometric determination of traces of cadmium(II) and copper(II) with 2-(5-bromo-2-pyridylazo)-5-diethylaminophenol in the presence of polyglycol octylphenyl ether. Microchem J 36: 386-390.
- CB Ojeda, AG de Torres, FS Rojas, JM Cano Pavon (1987) Determination of copper with 1, 5-bis (di-2-pyridylmethylene) thiocarbonohydrazide: Determination of copper in alloys. Microchem J 35: 164-167.
- S Li, S Li, A Chen (1993) Spectrophotometric determination of trace copper with a Cu-diethyldithiocarbamate-ß-cyclodextrin colour system. Talanta 40: 1085-1090.
- Dalman, M Tufekci, S Nohut, S Guner, S Karabocek (2002) Spectrophotometric determination of copper in pharmaceutical and biological samples with 3-{2-[2-(2-hydroxyimino-1-methyl-propylidene-amino)-ethylamino]-ethyl-imino}-butan-2-one oxime. J Pharm Biomed Anal 27: 183-187.
- HM Ma, YX Huang, SC Liang (1996) A new polymeric chromogenic reagent for the determination of copper(II). Anal Chim Acta 334: 213-219.
- IS Balogh, M Ruschak, V Andruch, Y Bazel (2008) An investigation of the reaction of copper ions with dimethylindodicarbocyanine dye. An application for the determination of Cu(I), Cu(II) and Cu(III). Talanta 76: 111-115.
- M Thakur, MK Deb (1999) The use of 1-[pyridyl-(2)-azo]-naphthol-(2) in the presence of TX-100 and N,N'-diphenylbenzamidine for the spectrophotometric determination of copper in real samples. Talanta 49: 561-569.
- S Nohut, S Karabocek, S Guner, Y Gok (1999) Extraction and spectrophotometric determination of copper(II) with S,S-bis(2-aminophenyl) oxalate. J Pharm Biomed Anal 20: 309-314.
- D Fu, D Yuan (2007) Spectrophotometric determination of trace copper in water samples with thiomichlersketone. Spectrochim Acta (A) 66: 434-437.
- R Chaisuksant, W Palkawong-naayuthaya, K Grudpan (2000) Spectrophotometric determination of copper in alloys using naphthazarin. Talanta 53: 579-585.
- L Yuan, SH Huo, XN Ren, H Chen Chin (2008) A novel spectrophotometric determination of copper(II) with bromosulphonazo III. Chem Lett 19: 92-94.
- KH Reddy, NBL Prasad, TS Reddy (2003) Analytical properties of 1-phenyl-1,2-propanedione-2-oxime thiosemicarbazone: simultaneous spectrophotometric determination of copper(II) and nickel(II) in edible oils and seeds. Talanta 59: 425-433.
- JN Miller, JC Miller (2005) Statistics and chemometrics for analytical chemistry. (5th edn), Prentice Hall, England.
Author Details
Ragab Y Sharaf1, Waheed M Salem1, Alaa E Ali1 and Alaa S Amin2*
1Chemistry Department, Faculty of Science, Damanhor University, Egypt
2Chemistry Department, Faculty of Science, Benha University, Egypt
Corresponding author
Alaa S Amin, Chemistry Department, Faculty of Science, Benha University, Benha, Egypt.
Accepted: October 31, 2020 | Published Online: November 02, 2020
Citation: Sharaf RY, Salem WM, Ali AE, Amin AS (2020) Utilization of Aluminon for Determination of Ultra-trace Levels of Copper in Environmental, Biological, and Water Samples via Cloud Point Extraction Coupled with Spectrophotometry. Int J Analyt Bioanalyt Methods 2:013.
Copyright: © 2020 Sharaf RY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The present paper outlines novel, simple and sensitive method for the determination of copper using spectrophotometry after separation and preconcentration by cloud point extraction. The cloud point methodology was successfully applied for copper determination after complexation with aluminon (triammonium salt of aurintricarboxylic acid), at pH 3.5. Copper(II) ions was quantitatively recovered in Triton X-114 after centrifugation. Acetonitrile (0.2 ml) was added to the surfactant-rich phase prior to its analysis at λmax 538 nm. The influence of analytical factors including complexing agent, buffer solutions, Triton X-114, temperature, heating time, centrifuge rate and time were examined and optimized. The analytical characteristics of the method (e.g. linear range, Ringbom concentration ranges, molar absorptivity, Sandell sensitivity, optimum limits of detection and quantification, enrichment factor, and improvement factors) were evaluated. Linearity was obeyed in the range of 7.5-220 ng/ml of Cu2+ ion. The detection and quantification limits of the procedure were 2.2 and 7.4 ng/ml of Cu2+ ion. The tolerance limits of some anions and cations were also examined. The procedure was applied to detect copper in environmental, water and biological samples.
Keywords
Copper determination, Cloud point extraction, Triton X-114, Complexation, Colorimetry, Environmental and biological analysis
Introduction
In biological materials, particularly biological fluids, might be considered a difficult analytical task, mostly because the complexity of the matrix and the low concentration of these elements, which requires sensitive instrumental techniques and often a preconcentration step. Monitoring the presence of toxic trace elements in biological fluids is an extremely essential task to establish occupational and environmental exposure [1-3]. Copper is an important micronutrient and plays an essential role in lipid metabolism and carbohydrate, while at high levels it is an environmental pollutant [4-6].
Copper plays a vital role in the biological systems. In biological systems, copper counter acts the toxicity of zinc suggesting Cu\\Zn antagonism [7]. In general, a daily copper intake of 1.5-2.0 mg is essential. However, copper becomes toxic to humans if a large amount is ingested and accumulated in the tissue. For example, because of its mobilization and redox activity, cata¬lytic copper is believed to play an important role in the formation of reactive oxygen species (ROS), such as superoxide anion (O2-) and OH radicals. These radicals bind very quickly to DNA and create damage by breaking the DNA strands or modifying the bases and/or deoxyribose, leading to carcinogenesis [8]. Overexposure to copper causes ptyalism, nausea, vomiting, epigastric burning, and diarrhea. High doses of copper result in a series of systematic toxic effects such as hemolysis, hepatic neurosis, gastrointestinal bleeding, oliguria azotemia, hemoglobinuria, hematuria, proteinuria, hypertension, tachycardia, convulsions, and coma [9]. Humans are commonly exposed to copper from drinking water, breathing air, foods or through having skin contact with copper, particles attached to copper or copper-containing compounds [10].
The determination of ultra-trace copper in biological samples is particularly difficult because of the matrix complexity and the usually low concentration of copper, which needs high sensitive instrumental techniques and frequently a preconcentration step [11-13]. However, an excessive uptake of Cu2+ can cause serious health problems, including ischemic heart disease, kidney disease, neurodegenerative disease, anemia and bone disorders [14]. Due to their toxicity, the maximum contamination value of Cu2+ in the environment and in food was set by several organizations throughout the world to protect human health. For example, the United States Environmental Protection Agency (USEPA) issued the maximum contamination of Cu2+ in drinking water at 1.30 mg/L [15]. In Thailand, the pollutant control organization permitted a Cu2+ level of 2.00 mg/L in surface water [16]. In addition, the concentration limit of Cu2+ for exposure from foods is in the range of 1.2-4.2 mg Cu2+/day as set by the European Food Safety Authority (EFSA) [17]. Therefore, the monitoring of ultra-trace levels Cu2+ contaminants in water, food and the environment is necessary.
Recently, various nanocomposites were used as sonochemical fabrication and demonstration as a novel compoundes for removal of organic and inorganic materials [18-22]. A variety of analytical advanced procedures requiring high priced instrumentation are recommended to be dealing with the detection of Cu2+ from different matrices at trace amount. These procedures include: Flame atomic absorption spectrometry (FAAS) [23], graphite furnace atomic absorption spectrophotometry (GF-AAS) [24], inductively coupled plasma emission spectrophotometry (ICP-ES) [25]. Although these analytical techniques are highly sensitive and selective, they are quite expensive for the developing countries and need well qualified laboratory personnel and superior laboratory equipment. Moreover, they might not be always available in all routine analytical laboratories. Various of the recommended procedures have some short comings like the availability of sophisticated instruments, tedious work, multi-step procedures for pre-concentration of the analyte, time-consuming operation and low selectivity with higher interference problems. The reported procedures [23] have demerits with respect to their high cost and the instruments used in day-to-day analysis.
Cloud point extraction (CPE) is depended on the phase character of non-ionic surfactants in aqueous solutions, which exhibit phase separation after increasing temperature or addition of a salting-out agent. Separation and preconcentration based on CPE are becoming an essential and practical application of surfactants in analytical chemistry [26,27]. The technique is based on the property of most nonionic surfactants in aqueous solutions to form micelles and to separate into a surfactant-rich phase of a small volume and a diluted aqueous phase when heated to a temperature known as the cloud point temperature. The small volume of the surfactant-rich phase achieved with this technique permits the design of extraction schemes that are cheap, simple, speedy, highly efficient, and of lower toxicity to the environment than those extractions that use the organic solvents. CPE might be an interesting and efficient alternative, once it reduces or eliminates consumption of organic solvents significantly. Trace elements can be extracted to the surfactant-rich phase usually after forming of a hydrophobic complex with an appropriate complexing agent [28]. This approach has been successfully employed to extract and preconcentrate several trace elements from a variety of matrices [28-30] including biological samples [31-34]. Any analyte solubilized in the hydrophobic core of the micelles will be concentrated into the small volume of the surfactant rich phase which can subsequently be detected by various spectrometric techniques such as FAAS [35], electrothermal atomic absorption spectrometry (ETAAS) [36], inductively coupled plasma mass spectrometry (ICP-MS) [37], inductively coupled plasma optical emission spectrometry (ICPOES) [38], laser induced-thermal lens spectrometry (LITLS) [39], and spectrophotometry [40].
The aim of this article was to extend the use of cloud point pre-concentration strategy to determine copper in various samples for the first time by using triammonium salt of aurintricarboxylic acid (aluminon) as complexing agent and Triton X-114 as surfactant coupled with colorimetry. Triton X-114 was chosen as nonionic surfactant because of its wide use as a cloud point surfactant as well as its low price, commercial availability and lower toxicity.
Material and Methods
Chemicals and reagents
Analytical-grade cupric nitrate, mercuric chloride, sodium molybdate, sodium tungstate, sodium acetate, hydroxylamine hydrochloride and nitrate salts of sodium, potassium, lithium, calcium, barium, beryllium, magnesium, manganese, strontium, cobalt, iron, lead, and nickel (brought from Merck) were of the highest purity available and used without any further purification. Double distilled water was used throughout.
High-purity ethanol, methanol, isopentyl alcohol, chloroform, nitric acid, hydrochloric acid, and ammonia solution (all from Merck) were used without any further purification.
Preparation of cooper and aluminon stock solutions
A standard stock copper solution (1000 mg/L) was prepared by dissolving 0.3802 g of Cu(NO3)2•3H2O in double distilled water and diluting to 100 ml in a calibrated 100 ml flask. This solution was standardized by iodimetric titration. Working solution were prepared by appropriate dilution of the stock solution. A 5.0% (v/v) Triton X-114 from E. Merck, Darmstadt, Germany was prepared by dissolving 5.0 ml of Triton X-114 in double distilled water in 100 ml volumetric flask with stirring. Cloud points of Triton X-114 in aqueous solution are 24 ℃ [41].
A stock solution of 5 × 10-3 M aluminon (achieved by Merck) was prepared by dissolving an appropriate amount in least volume of double distilled water and completed to the mark in 50 mL measuring flask. Acetate, borate, phosphate, thiel and universal buffer solutions of different pH values 2.0-12 were prepared as described early [42]. Stock solutions of 2000 μg/ml of interfering ions were prepared by dissolving appropriate amounts of suitable salts in double distilled water.
Apparatus
All Absorption spectra and absorbance measurements were recorded and measured with a Perkin-Elmer λ3B double beam UV-VIS spectrophotometer with 0.5 mm quartz cuvettes. An Orion research model 601 A/digital ionalyzer fitted with a combined glass-calomel electrode was used for pH adjustment and checking the pH value of buffer solutions. An inductively coupled plasma (ICP) model Varian Liberty 150AX Turbo was used for copper concentration detection. A 30E 148 Sheme fan or Hettich centrifuge was used to accelerate the phase separation process. A water bath with a good temperature control within ± 2.0 ℃ and a centrifuge with 10 ml calibrated centrifuge tubes (Superior, Germany) were used to achieve and accelerate the phase separation process, respectively.
General procedure
For the cloud point extraction, 5.0 ml of pH 3.5 thiel buffer solution, 4.0 ml of 5 × 10-3 M aluminon solution and 3.0 ml of 5.0% (v/v) Triton X-114 solution, and an aliquot of 10 ml of a solution containing Cu2+ ion (0.75-22.0 μg), were kept for 5.0 min in the thermostatic bath at 50 ℃ and then completed to 100 ml in a 100 ml measuring flask. Separation of two phases was achieved by centrifugation for 5.0 min at 4000 rpm. The mixture was cooled in an ice bath to increase the viscosity of the surfactant-rich phase, and the aqueous phase was easily decanted by simply inverting the tube. The micellar extract from this procedure was diluted with 0.2 ml of acetonitrile and transferred into a 0.5 ml quartz cell to record its absorbance at 538 nm.
Pretreatment of samples
Analysis of water (tap, Waste and river) samples for determination of copper ions content was performed as following: 400 ml of sample was poured in a beaker and 8.0 mL concentrated HNO3 and 3.0 ml of 30% (v/v) H2O2 for elimination and decomposition of organic compound were added. The samples, while stirring was heated to one tenth volume. After adjustment of samples pH to desired value, the complexation and CPE were performed according to described general procedure.
Homogenized soil sample 20g or human blood from Health individuals sample (20 ml) was accurately taken and in a 200 ml beaker was digested in the presence of an oxidizing agent with addition of 10 ml concentrated HNO3 and 2.0 ml 70% of HClO4 was added and heated for 1.0 h. The content of beaker was filtered through a Whatman No. 40 filter paper into a 250 ml measuring flask and its pH was adjusted to desired value and diluted to mark with bi-distilled water. Then the above general procedure was applied. Copper levels in the final solutions were determined by ICP-AES and the above general procedures.
Spinach and lotus samples were purchased from Benha, Egypt. Afterwards, it was taken in small mesh. For the digestion of the sample, the procedure given by Ghaedi [43,44] was applied. A 40g of spinach and lotus samples were heated in silica crucible for 3.0 h on a hot plate and the charred material was transferred to furnace for overnight heating at 650 ℃. The residue was cooled, treated with 10 ml concentrated nitric acid and 3.0 ml 30% H2O2 again kept in furnace for 2.0 h at the same temperature so that no organic compound traces are left. The final residue was treated with 3.0 ml concentrated hydrochloric acid and 2.0-4.0 ml 70% perchloric acid and evaporated to fumes, so that all the metals change to respective ions. The solid residue was dissolved in water, filtered and by keeping the pH at 4.5 made up to 25 ml by addition of thiel buffer. The dissolved solution was suitably diluted and presented procedure was applied.
A 50g of goat liver and/or meat were taken and dried for 48 h in an oven at 120 ℃ to remove the water content and to obtain a constant weight (about 68% water). Dried goat liver and/or meat sample were transferred into a glass flask. For the digestion of the sample, the procedure given by Ghaedi, et al. [43] was applied and the samples were treated according to previous publication [45,46]. A concentrated acid mixture of 3.0 ml H2SO4, 15 ml HClO4, and 15 ml HNO3 was added and left to stand over night. The solution was kept in an oil bath at 50 ℃ until the foaming stopped. Then the temperature was increased to 150 ℃ and heating was continued until the evolution of brown fumes of nitrogen oxides ceased. When a dark brown in mixture was appeared, the flask was cooled for about 2.0 min then a 5.0 mL of nitric acid had to be added. Heating was continued until nitrogen oxides fumes were longer given off. Appearance of white fume of perchloric acid in 1.0 ml solution is an indication of complete digestion. The solid residue was dissolved in water and filtered and by keeping the pH at 4.5 made up to 25 ml by addition of thiel buffer. Then the procedure was applied. Copper levels in the final solutions were determined by ICP-AES and the above general procedures.
Results and Discussion
Aluminon represents maximum absorbance at 336 nm at pH 3.5. Cu(II) in buffer medium of pH 3.5 reacts with aluminon in absence (λmax 518 nm) and in the presence of Triton X-114, the absorbance of solution presented a bathochromic shift to 538 nm. Therefore the ternary complex of Cu2+- aluminon - Triton X-114 can be extracted by CPE method. After separation of surfactant-rich phase, the absorbance was measured at 538 nm against a reagent blank as the reference (Figure 1). Therefore, the method is suitable for preconcentration and spectrophotometric determination of copper.
Optimization of the system
To take full advantage, the reagent concentrations and reaction conditions must be examined and optimized. Various experimental factors were illustrated in order to achieve optimization. These factors were optimized by setting all parameters to be constant and optimizing one each time.
The formation of Cu2+-complex and its chemical stability are the two important influence parameters for CPE. The pH plays a unique role on Cu2+-complex formation and subsequent extraction, and is proved to be a main factor for CPE. Extraction yield depends on the pH at which complex formation is carried out. The influence of pH on the absorbance at a constant concentration of complex in surfactant-rich phase was established in the range of 2.5-11.5. Various types of buffers (acetate, borate, phosphate, thiel, and universal) [42] were tested. The optimum one was the universal buffer, and the best values were between pH 3.0-4.0 (Figure 2). As Figure 2 shows, the pH 3.5 was selected for all further investigates, since the results is higher concordant at this value. The complexation reaction at pH values lower than 2.75 is incomplete due to protonation of reagent and complexation reaction is incomplete. The decrease in absorbance at pH values higher than 8.0 could be due to the hydrolysis of Cu2+. Moreover the optimum volume of pH 3.5 per 100 ml sample was found to be 5.0 ml, since the results is highly concordance at this volume.
The influence of concentration of the complexing agent on the analytical responses was subsequently illustrated. The effect of aluminon concentration on the extraction and detection of Cu2+ was established in the range (0.5-5.0) × 10-4 M. The sensitivity of the procedure increased by increasing aluminon concentration up to 1.5 × 10-4 M and remained constant at higher concentrations. Therefore, 1.5 × 10-4 M of aluminon was applied in further works. The results are given in Figure 3.
It was desirable to employ the shortest equilibration time and the lowest possible equilibration temperature as a compromise between completion of extraction and efficient separation of phases. The dependence of extraction efficiency upon equilibration temperature and time above the cloud point in the range of 30-60 ℃ (Figure 4), and 5.0-25 min, were thoroughly optimized, respectively. The results represented that an equilibration temperature of 50 ℃ and an equilibration time of 5.0 min were adequate to achieve quantitative extraction. It was found that 50 ℃ is adequate for these analyses. An equilibration time of 5.0 min was selected as the optimal to reach maximum sensitivity. Unreasonably high temperatures are not suitable in the proposed analytical procedure since they could create stability problems for complexes and complexing agents, while at higher temperature, CMC of non-ionic surfactants decreases [47]. The results indicate the experiment in the optimized reagent concentration after heating for 5.0 min at 50 ℃ and centrifuging by 5.0 min in 4000 rpm and cooling in 5.0 min in ice-bath lead to high recovery of Cu2+ ion in short time. Moreover, non-ionic surfactants appear relatively more hydrophobic at higher temperatures, due to an equilibrium shift that favors dehydration of the ether oxygen's [48]. This leads to an increase in the micelles concentration. The solubilization capability of the micellar solution increases with temperature leading to an increase in the complex extraction [48].
The concentration of surfactant that is used in CPE is critical parameter. To obtain the optimal concentration of Triton X-114, the influence of surfactant concentration on the increase in the absorbance of the system was established in the range 0.025-0.25% (v/v). As Figure 5 represents, the absorbance of the solutions increased by increasing the Triton X-114 concentration up to 0.15% (v/v) and remained constant at higher concentrations. The increase in absorbance by increasing surfactant concentration can be due to both more efficient extraction of the complex and medium effect. Therefore, 0.15% (v/v) Triton X-114 was used in further investigates.
As the surfactant-rich phase was very viscous, acetonitrile was added to the surfactant-rich phase after CPE to facilitate its transfer into spectrophotometric cell. The amount of 0.2 ml acetonitrile was chosen to have an appropriate amount of sample for transferring and recording the sample absorbance. Therefore, a preconcentration factor of 500 was obtained.
Stoichiometric ratio
The nature of the complex was illustrated at the optimum conditions described above using the molar ratio and continuous variation methods. The plot of absorbance versus the molar ratio of aluminon to Cu2+, obtained by varying the aluminon concentration, showed inflection at molar ratio 2.0 (Figure 6), indicating presence of two aluminon molecules in the formed complex. Moreover, the Job method represented a ratio of aluminon to Cu2+ = 2.0. Consequently, the results showed that the stoichiometric ratio was (2:1) [aluminon:Cu2+]. The conditional formation constant (log K), evaluated using Harvey and Manning equation applying the data obtained from the above two methods, was brought to be 4.37, whereas the true constant was 4.48.
For ternary complexes of Cu- aluminon -Triton X-114, the stoichiometric ratio as achieved from molar ratio represented the formation of 1:1 for [Cu(aluminon)2] : Triton X-114; so we conjectured that ternary complex [Cu(aluminon)2] [Triton X-114] is formed in the system. The structure of which is probably as follows:
Cu2+ + 2 aluminon [Cu(aluminon)2]
[Cu(aluminon)2] + [Triton X-114] {[Cu(aluminon)2][Triton X-114]}
Calibration curve and sensitivity
The calibration curve showed that the system obeys Beer's law in the concentration range of 7.5-220 ng Cu2+ per ml in the measured solution (working range). For more accurate results, Ringbom optimum concentration ranges was obtained by plotting transmittance percentage against the logarithmic value of Cu2+ concentration and found to be 25-205 ng Cu2+per ml in the measured solution. The linear regression equation obtained was A = 3.02 C (μg/ml) + 0.005 (r = 0.9996). The molar absorptivity was evaluated to be 1.92 × 107 L/mol.cm at 538 nm, whereas Sandell sensitivity was found as 0.0033 ng/cm2. Because the amount of Cu2+ in 100 ml of sample solution is measured after preconcentration by CPE in a final volume of 0.2 ml, the maximum Enrichment factor of the solution is 500. The improvement factor, defined as the ratio of the slope of the calibration graph for the CPE procedure to that of the calibration graph in aqueous media, for Cu2+ was 1165 (Table 1).
The standard deviations of the absorbance measurements were calculated from a series of 13 blank solution. The limits of detection (K = 3) and of quantification (K = 10) of the method were established [49] and recorded in Table 1, according to the IUPAC definitions (C1 = KSo/s where C1 is the limit of detection, So is the standard error of blank, s is the slope of the standard curve and K is the constant related to the confidence interval). The relative standard deviation was 1.85% obtained from a series of 10 standards each containing 125 ng/ml of Cu2+.
The separation occurred efficiently, resulting in good enrichment factor and low LOD. In contrast to some other preconcentration techniques, no organic solvent is employed; therefore, the environmental pollution is limited to a small amount of surfactant. The methodology offers a facile, sensitive, inexpensive and nonpolluting alternative to other preconcentration techniques. The method is relatively rapid as compared with previously reported procedures for the enrichment of analyte ions (Table 2). The detection limits of analytes are superior to those of preconcentration techniques [50-56].
The sensitivity expressed as molar absorptivity of the proposed method is compared with those of published spectrophotometric methods (Table 3). The proposed method is more sensitive than other methods [57-70], that based on spectrophotometry (Table 3).
Interference
In order to study the selective separation and detection of copper ions from its binary mixtures with diverse metal ions, an aliquot of aqueous solutions (100 mL) containing 125 ng Cu2+ and mg amounts of other cations was taken and the proposed procedure was followed. The tolerance limit was taken as the ratio of foreign ions to the Cu2+ ion determined that causes a ± 5.0% error in the absorbance value. The results are summarized in Table 4. The result shows that most common ions do not interfere with the determination suggesting the highly selectivity of the proposed procedure.
Analytical applications
In order to confirm the applicability of the proposed procedure, it has been used to determine nanogram amounts of Cu2+ in environmental, in biological and water samples. The reliability of the presented procedure was checked by spiking experiments and independent analysis. The results for this study are presented in Table 5. An ICP-AES method was applied as a reference method and the results are also shown in Table 5. The recovery of spiked samples is satisfactorily reasonable and was confirmed using standard addition method, which indicates the capability of the system in the determination of ions. A good agreement was achieved between the added and measured analyte amounts. The recovery values evaluated for the added standards were always higher than 95%, thus confirming the accuracy of the method and its independence from the matrix effects.
The performance of the proposed method was assessed by calculation of the t-value (for accuracy) and F-test (for precision) compared with ICP-AES method. The mean values were obtained in a Student's t- and F-tests at 95% confidence limits for five degrees of freedom [71]. The results represented that the calculated values (Table 5) did not exceed the theoretical values. A wider range of determination, higher accuracy, more stability and less time consuming, represents the advantage of the proposed over other methods.
Conclusions
Cloud point extraction was applied to the preconcentration of copper in various samples. The procedure, which is based on the cloud point extraction of the complex of copper with aluminon, allows the determination of copper as low as 2.2 ng/ml. The proposed procedure needs inexpensive instrumentation and offers safety, good selectivity, accuracy and precision that can be used to the determination of copper in real samples. The surfactant has been applied for preconcentration of copper in samples, and thus toxic solvent extraction, has been avoided. The methodology offers a simple, rapid, inexpensive and nonpolluting alternative to other preconcentration techniques. The procedure is relatively rapid as compared with previously reported procedures for the enrichment of analyte ions.

