
International Journal of Optics and Photonic Engineering
(ISSN: 2631-5092)
Volume 7, Issue 2
Research Article
DOI: 10.35840/2631-5092/4551
Electron-Photon Interactions in Atomic Hydrogen: Significance of High (near zero) Energy States
Richard Sauerheber*
Table of Content
Figures
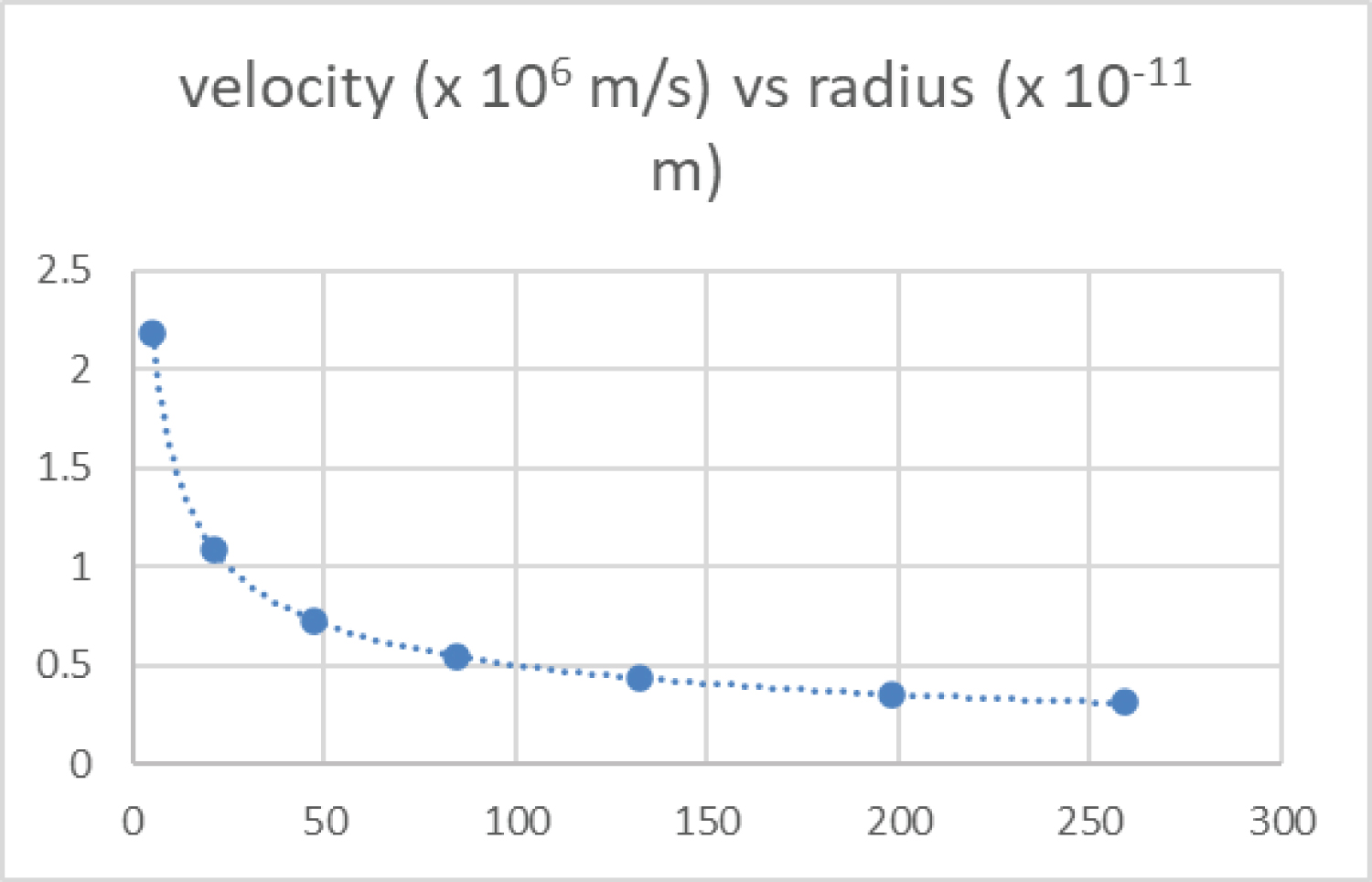
Figure 1: The most probable velocity...
The most probable velocity for the hydrogen atom electron is plotted as a function of the most probable radii of the orbits, progressively from n = 1 to 7 from Equations 1 and 3.
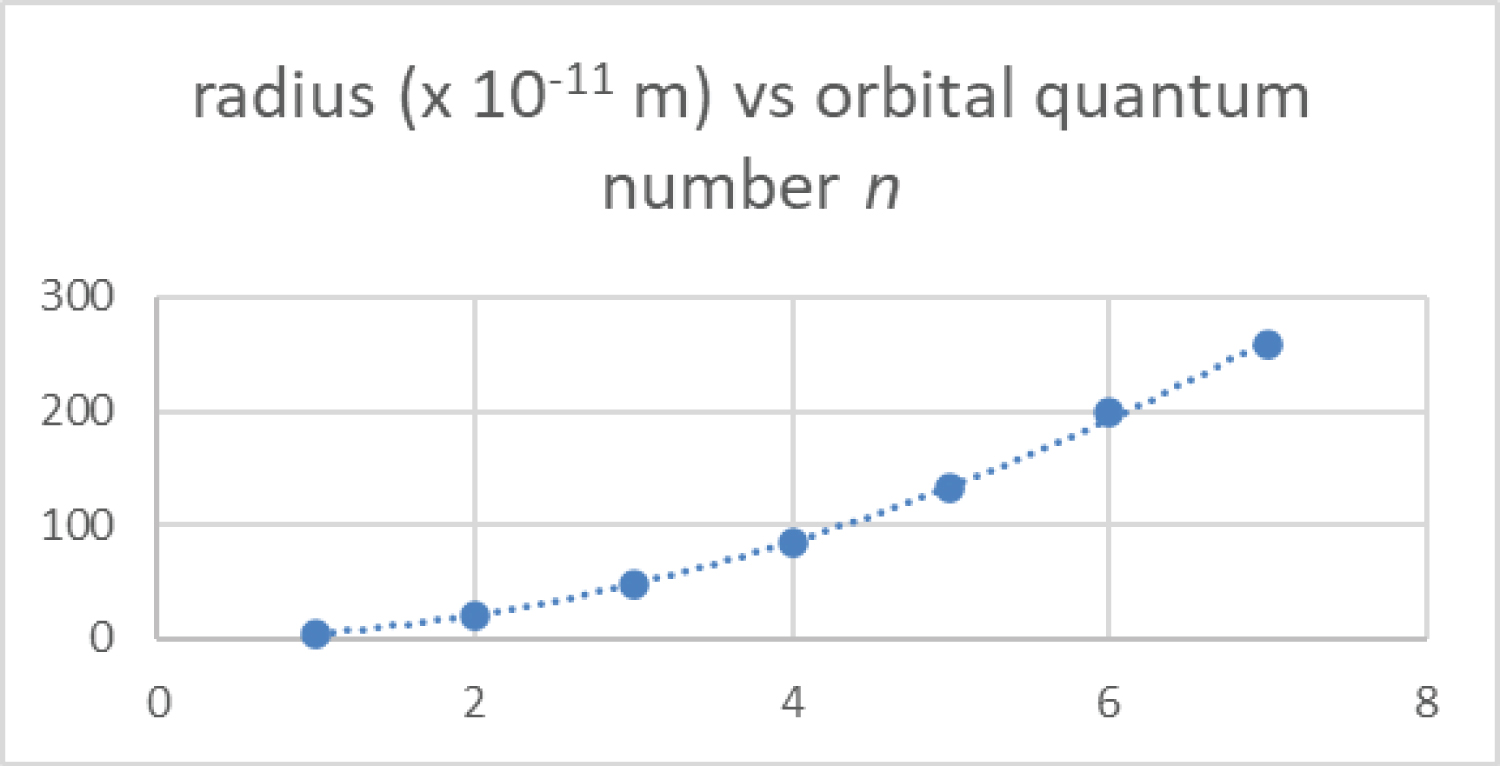
Figure 2: The calculated most....
The calculated most probable radius of the orbitals of the hydrogen electron are plotted as a function of principal quantum number from n = 1 to 7 from Equation 2.
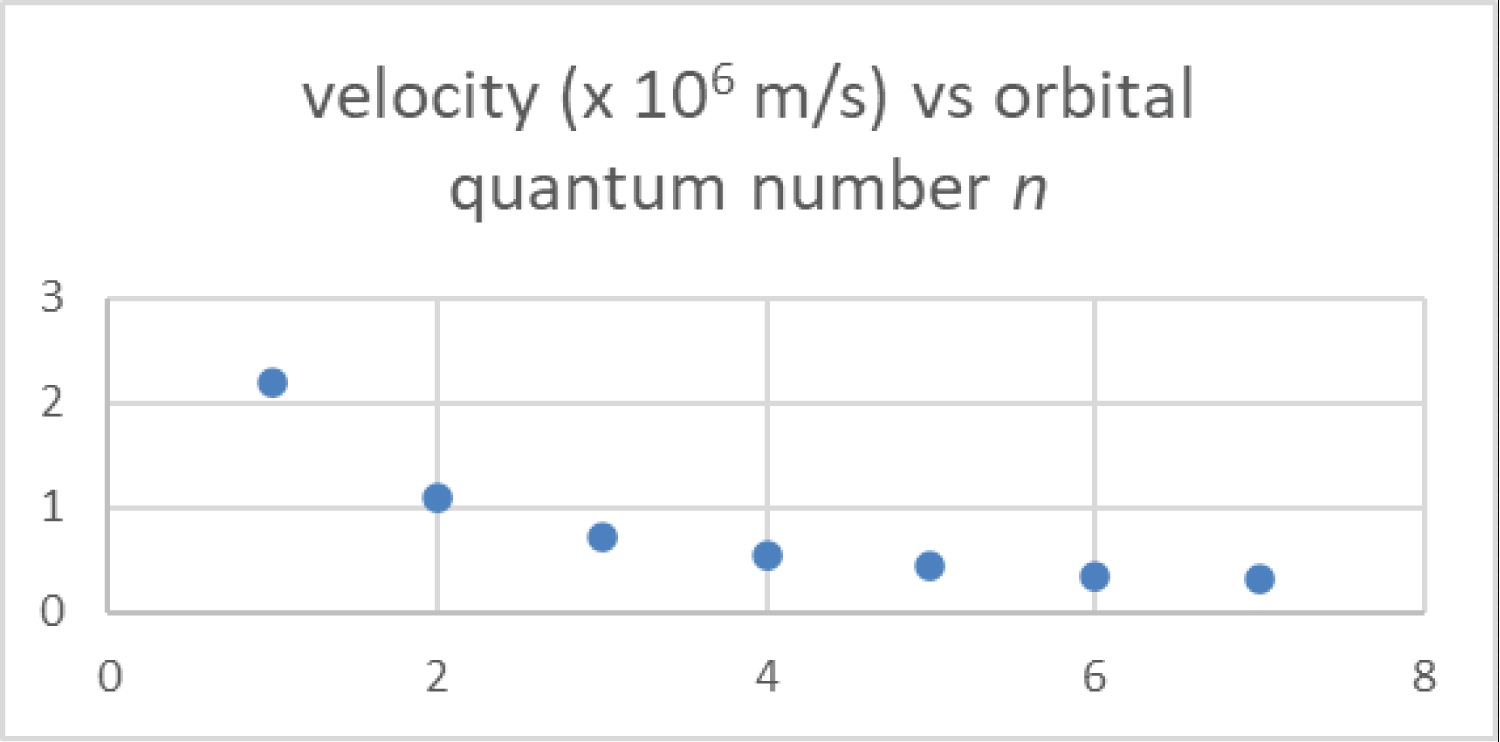
Figure 3: The velocity of the....
The velocity of the hydrogen electron is plotted as a function of the principal orbital quantum number for n = 1 to 7 from Equations 1 and 3.
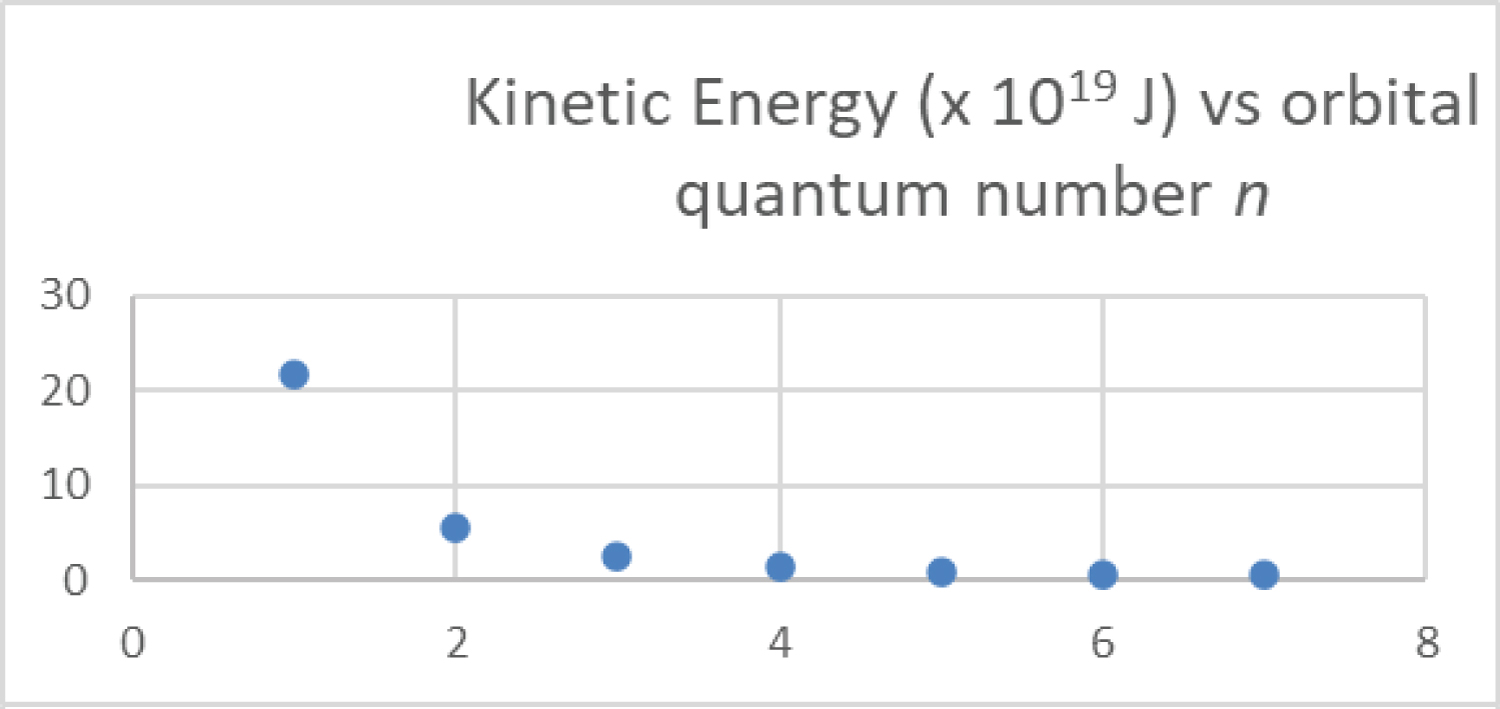
Figure 4: The most probable....
The most probable kinetic energy for the hydrogen electron is plotted as a function of the principal quantum number from n = 1 to 7 from Equation 3.
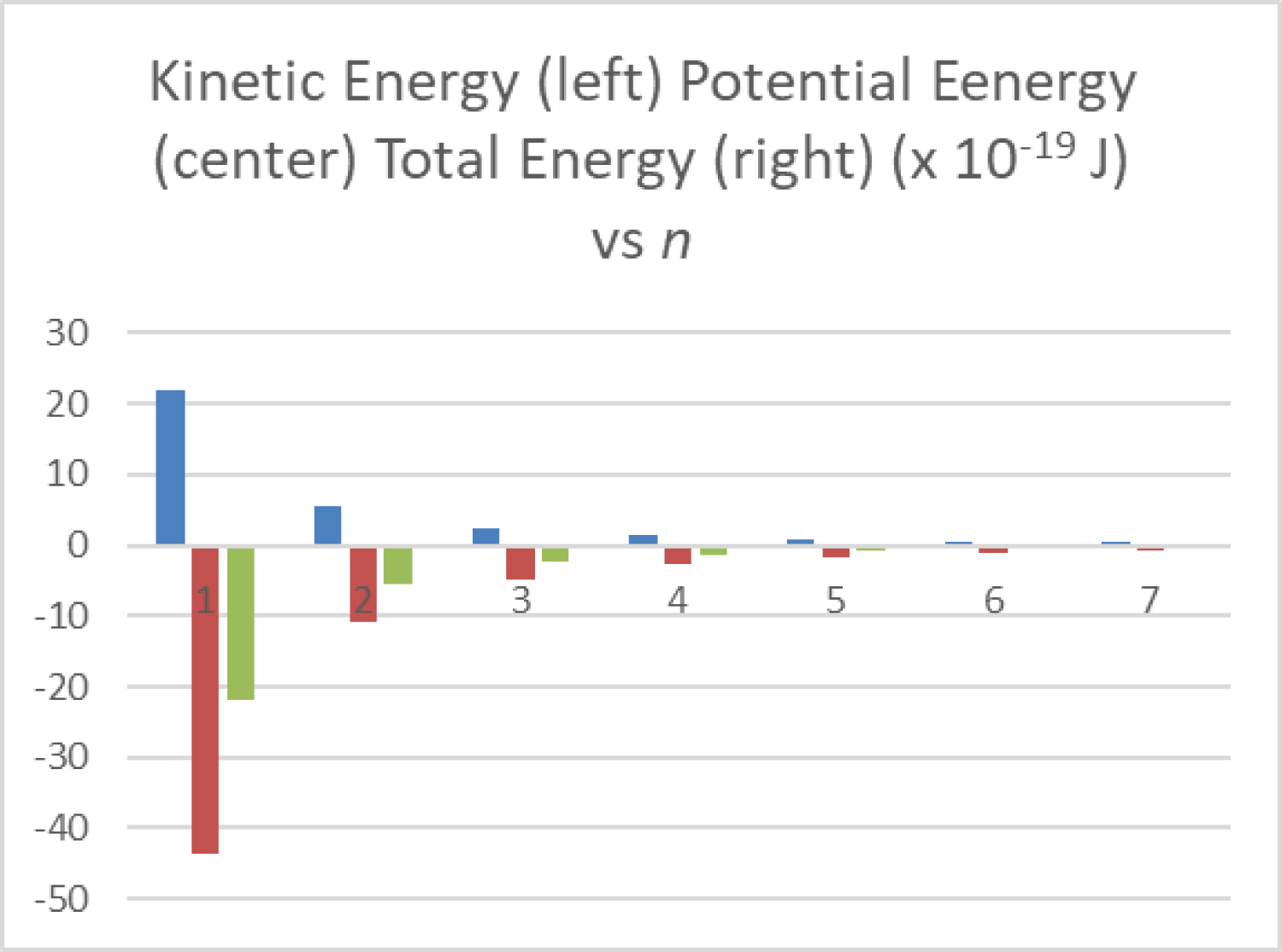
Figure 5: The kinetic energy....
The kinetic energy (left positive bars), potential energy (negative bars on right), and total orbital energy (negative bars at center) of the hydrogen electron are shown as a function of principle quantum number from n = 1 to 7.
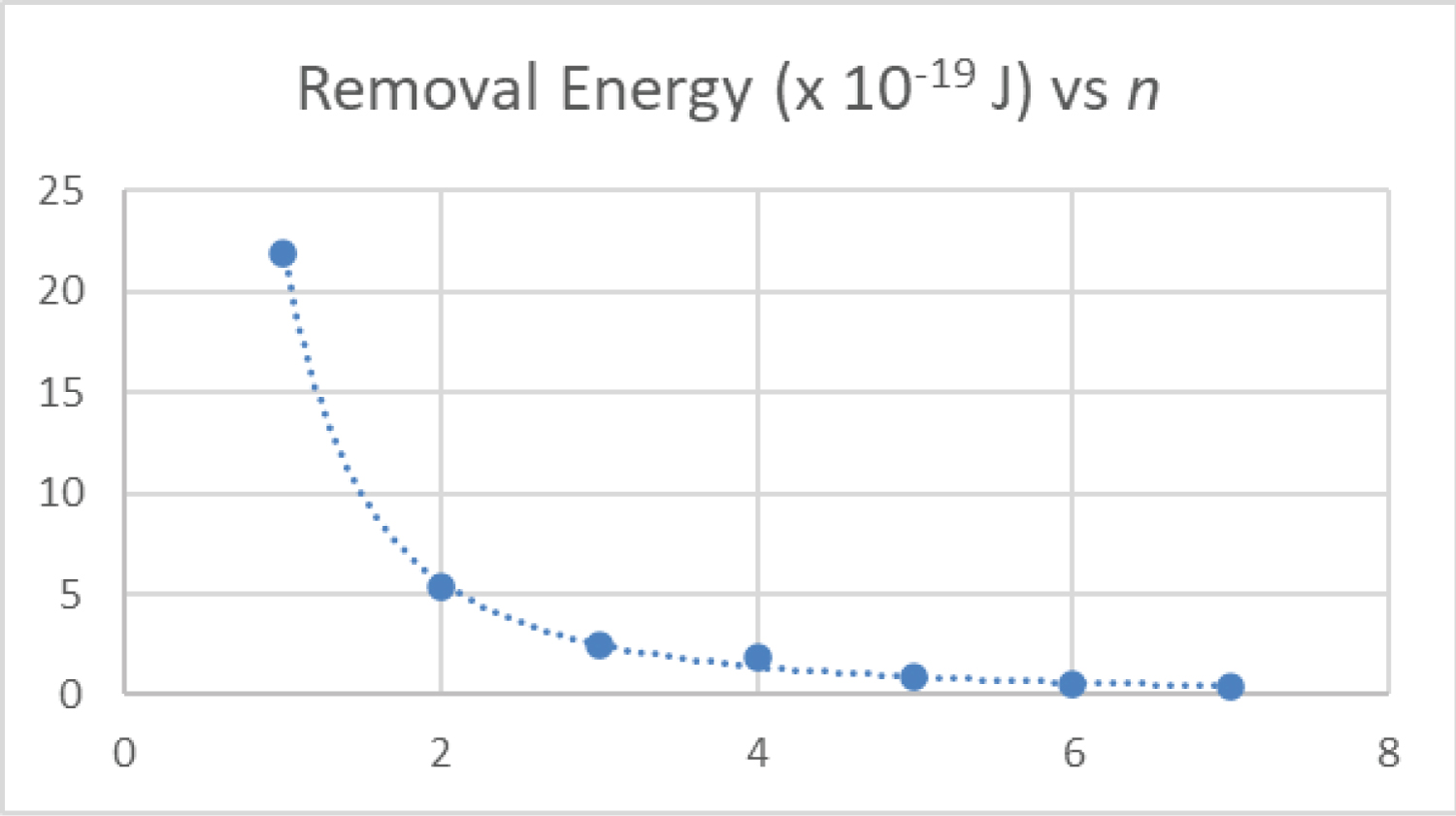
Figure 6: The energy computed....
The energy computed to remove the electron from the ground sate of hydrogen is plotted as a function of quantum number n.
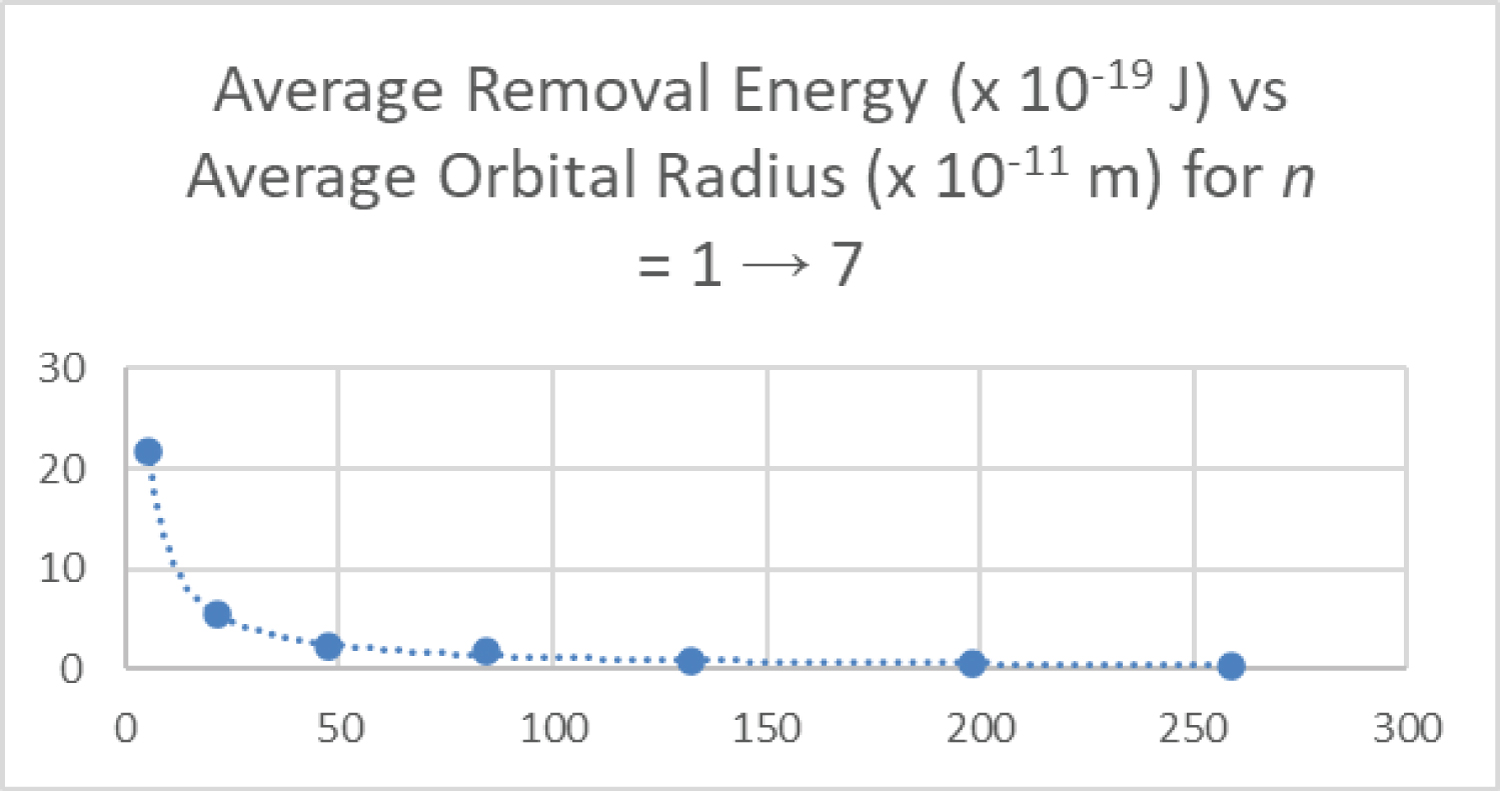
Figure 7: The ground state 1s...
The ground state 1s electron removal energy is plotted as a function of the most probable radius for the principle quantum number n from 1 to 7 for the hydrogen atom.
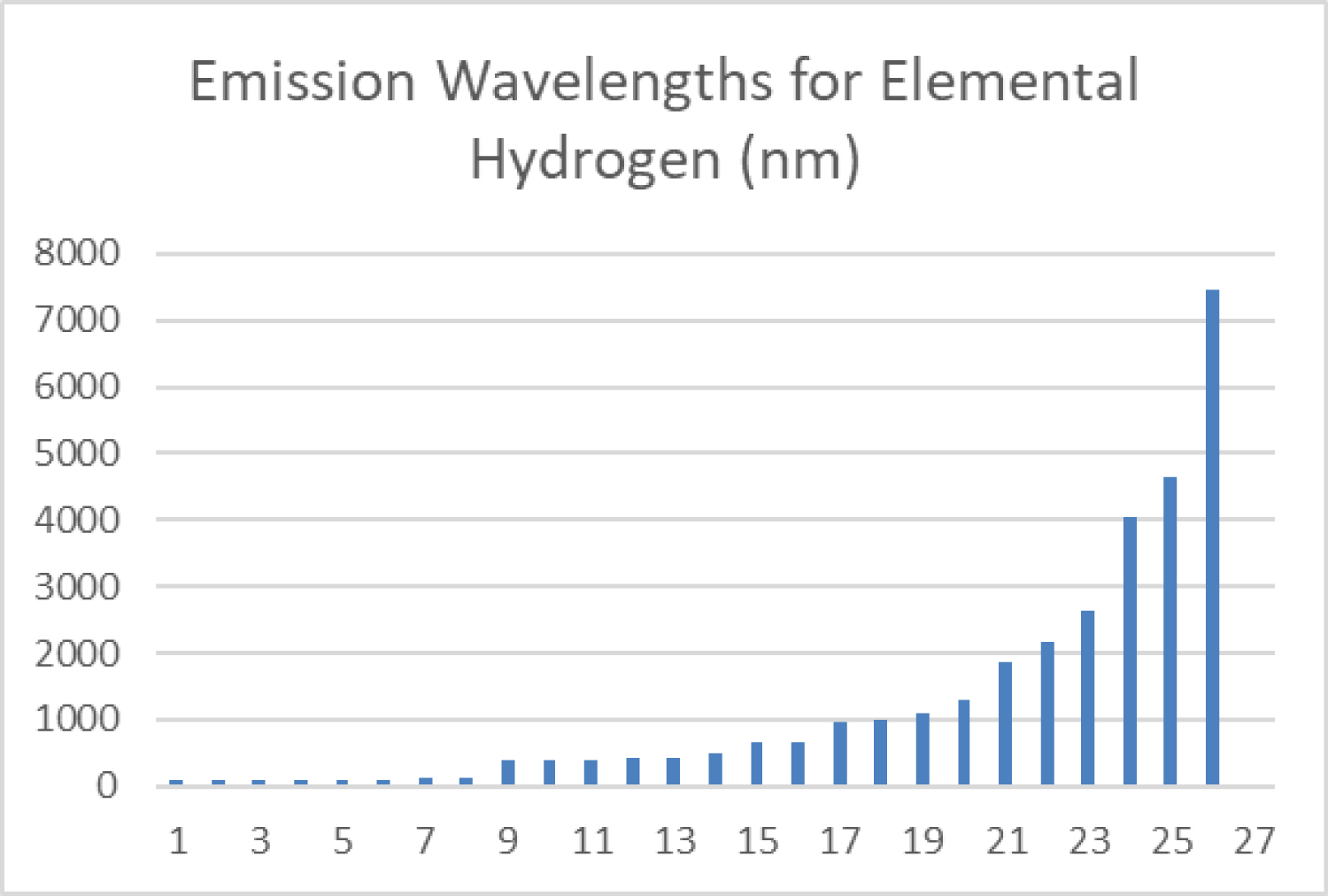
Figure 8: The spectroscopic wavelengths...
The spectroscopic wavelengths of light emitted by elemental hydrogen are shown in nanometer units from the lowest to the highest (from the highest energy transition to the lowest) for the first 26 distinctive emission lines of significant intensity. Data are from the CRC Handbook of Physics and Chemistry [4]. The first 8 reflect emissions from higher orbital quantum number to n = 1, the next 8 are from emissions to n = 2, the next 5 to n = 3, the next 3 are to n = 4, and the last two are to n = 5. Higher orbital transition emission lines are not reported because these are not distinguishable from each other, being of small energy differences between the higher adjacent orbitals. For example, the n = 8 to n = 1 transition emits a wavelength of 92.6 nm, and the n = 2 to n = 1 transition 122 nm (#8); from n = 5 to n = 2 is 434 nm and the 3 to 2 is 486 nm (#16); the 5 to 3 is 1282 and the 4 to 3 is 1876 nm (#21). The first 8 are to the 1s orbital, the only sublevel for n = 1, from various higher orbital quantum states. The next 6 are to the 2s orbital and #15 and #16 are to the 2p orbital. The next two are to the 3s and #21 and #22 are to the 3p and #23 is to the 3d, the three sublevels for the n = 3 quantum states. Less clear but by inference, the next two are to n = 4s and 4p, and the last two to n = 5s and 5p.
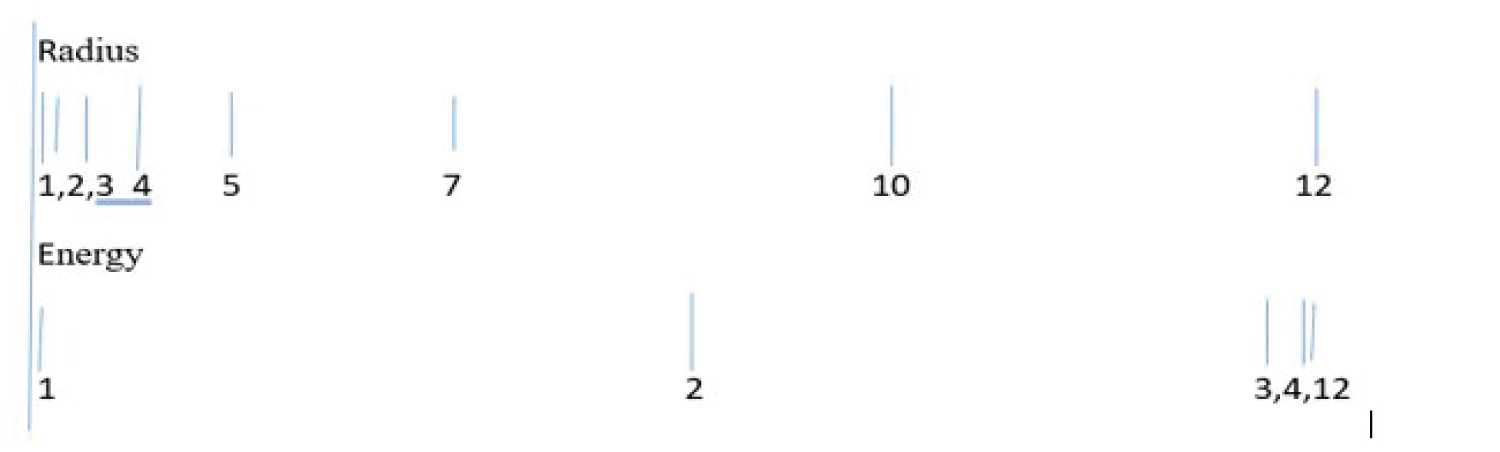
Figure 9: Differences in energy ...
Differences in energy (bottom row) and radius (upper row) are shown as a function of orbital quantum number, from n = 1 to 12. Energy differences between orbitals become small and tend toward zero as n increases, while radii for those orbitals of increasing n become very large.

Figure 10: Trends for average radius...
Trends for average radius A and orbital removal energy B for the hydrogen atom at large values of quantum number n. In the energized Rydberg state atom at n = 30, the orbital energy does not reach zero which is an untouchable asymptote, while the radius of the energized atom becomes huge.
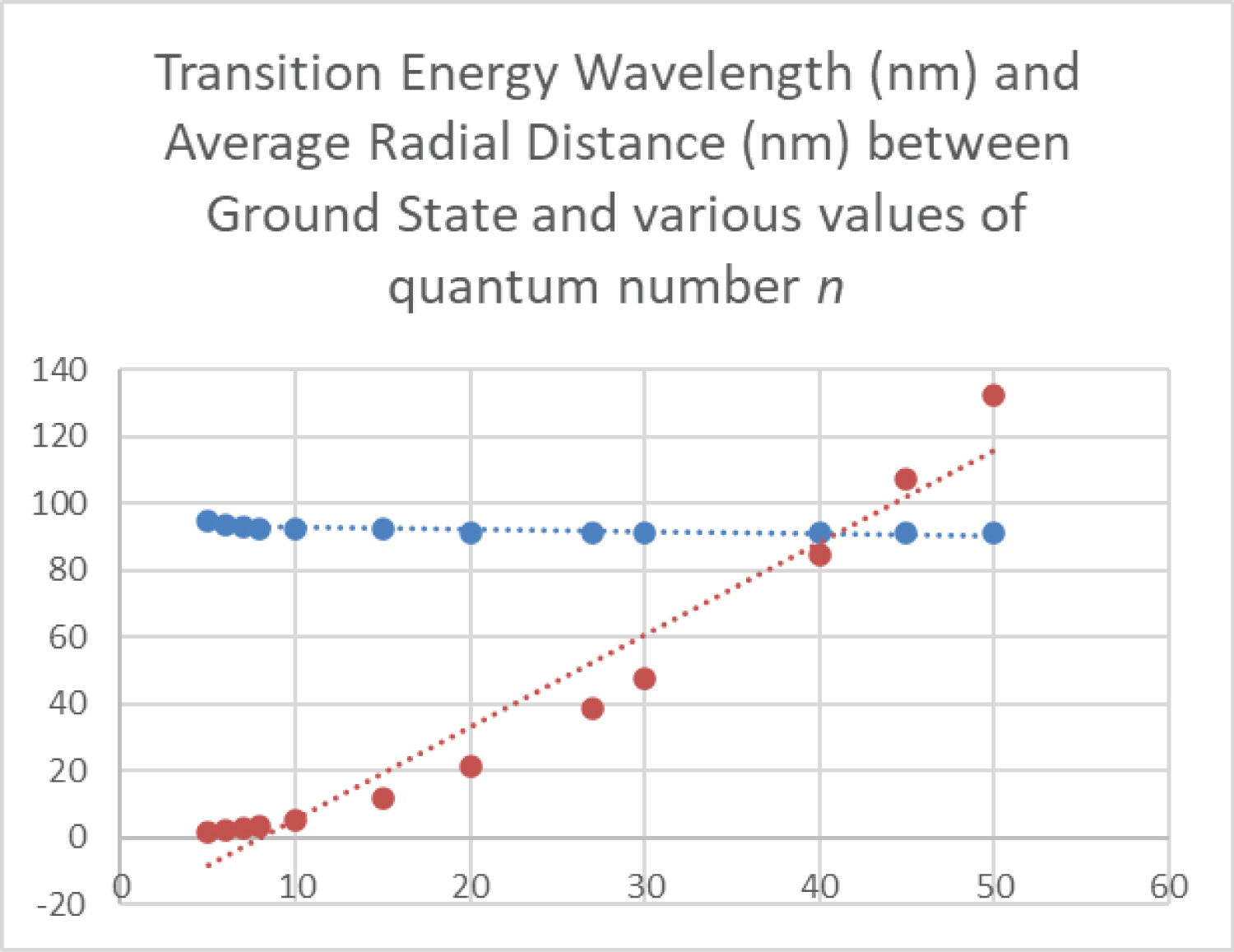
Figure 11: The wavelength (nm) of...
The wavelength (nm) of light required to induce a transition from the ground state to a higher energy state, and the radial distance (nm) between the ground state average radius and the average radius of that higher energy state are graphed as a function of principal quantum number n.
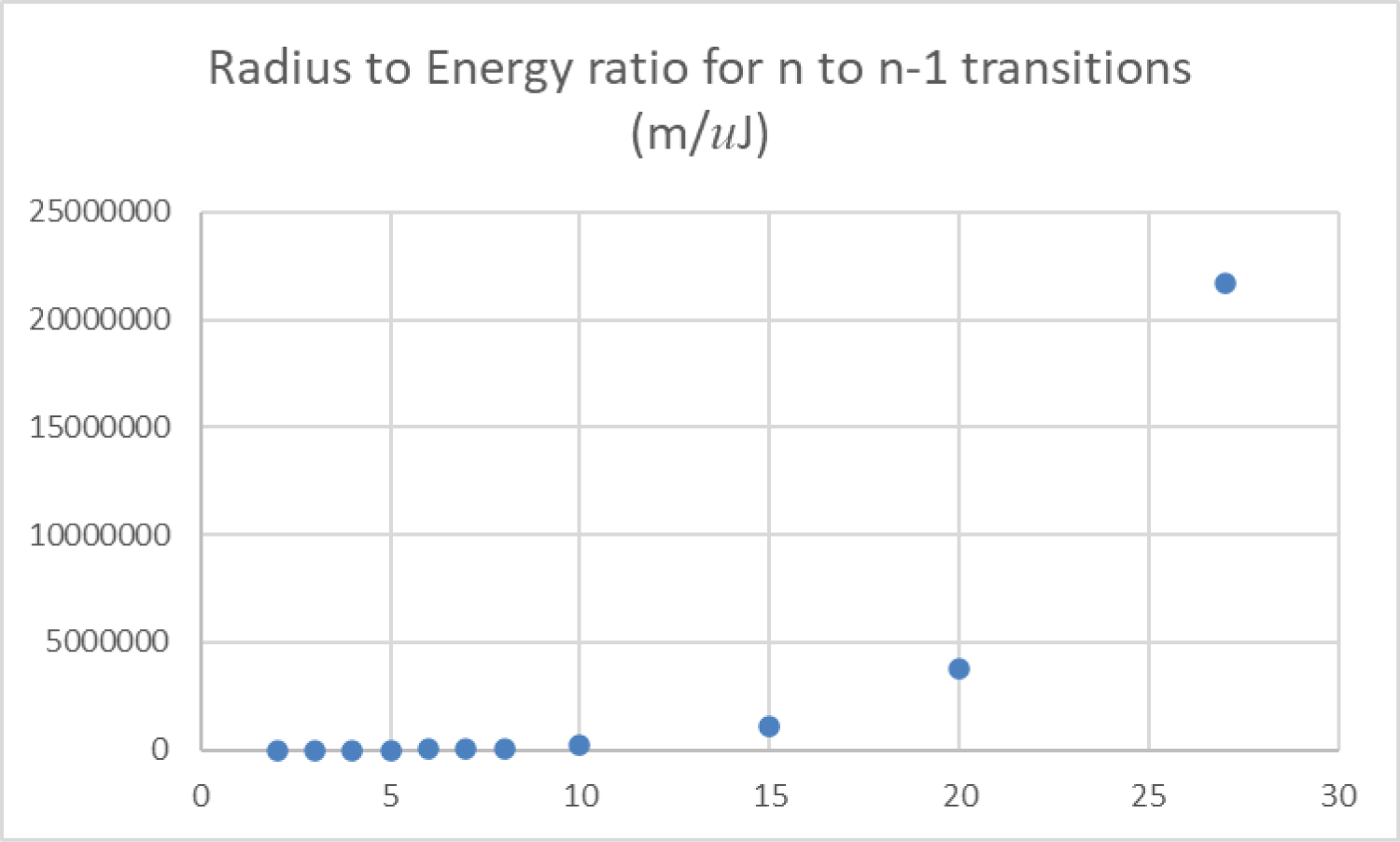
Figure 12: The distance between...
The distance between an orbital transition in meters from an orbital quantum number n to the adjacent lower orbital n-1 is plotted as a function of the energy required to induce that transition in micro-Joules.
Tables
Table 1: Summarizes calculations of the radii, velocities, kinetic and potential energies, and total energies for the hydrogen electron as a function of principal quantum number n.
References
- Cline D (2021) Virial Theorem. Physics Libre Texts 2.1.
- Lide DR (2007-2008) Handbook of Chemistry and Physics. CRC Press 1-6.
- McMurray J, Fay R, Robinson J (2016) Chemistry. (7 th edn), Pearson.
- Beiser A (1965) Concepts of modern physics. (6 th edn), McGraw-Hill, Inc., NY.
- NIST (2019) 2018 CODATA value: Rydberg constant. The NIST Reference on Constants, Units, and Uncertainty.
- https://www.sciencedirect.com/topics/chemistry/rydberg-state.
- Dunning FB, Killian TC (2021) Rydberg Atoms: Giants of the Atomic World, Scientia, Engineering and Tech Physical Science.
- Sauerheber R, Espinoza E (2018) Perspectives on solar system dynamics. Phys Astron Int J 2: 224-229.
Author Details
Richard Sauerheber*
STAR Center, Palomar Community College, USA
Corresponding author
Richard Sauerheber, STAR Center, Palomar Community College, 1140 W. Mission Rd., San Marcos, CA 92078, USA.
Accepted: November 03, 2022 | Published Online: November 05, 2022
Citation: Sauerheber R (2022) Electron-Photon Interactions in Atomic Hydrogen: Significance of High (near zero) Energy States. Int J Opt Photonic Eng 7:051.
Copyright: © 2022 Sauerheber R. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Orbital energies for the hydrogen atom are computed to elucidate kinetic and potential energy relative contributions to total energy. Formula from the Bohr quantum orbital energies and wave function treatments are compared to average energies derived from Newtonian mechanics. The difference between the theoretically calculated ground state energy and the highest energy photons of light that are absorbed by hydrogen in vacuum determines the residual energy of the electron that is usually considered ionized or labeled as "at infinity." Because the conversion of light energy into orbital energy is specifically quantized, the radial distance involved for a given required excitation energy appears to limit the maximum allowed electron transition. Newtonian mechanics helps explain characteristics of matter in the cosmos that are in common with electrons in atoms.
Keywords
Orbital energy, Potential and kinetic energy, Rydberg constant, High energy orbitals
Introduction
Light interaction with matter
Photons of light consist of electromagnetic energy contained in electric E and magnetic B fields that oscillate sinusoidally, tracing out wave patterns in space while propagating forward at fixed speed c = E/B in any particular medium, most rapidly in vacuum where light propagates in perpetuity when uninterrupted by matter. Light interacts with physical matter which can cause light to be reflected, refracted, scattered, or absorbed, depending on the structure of the material. When photon-electron interactions are associated with light absorption, the energy of the disappearing photon is conserved by causing the electron to be energized.
Absorption of a light photon within a narrow frequency range by chlorophyll in green plants causes ejection of an aromatic electron in the light-dependent reaction of photosynthesis. Absorption of a minimum frequency photon in conductive metals causes an electron to be energized in the photoelectric effect. In the case of absorption of light by elements in gas phase, a single photon of the proper frequency that matches the change in energy required to undergo a transition to a higher energy state is absorbed by a single electron. Unlike molecular and metallic absorption, energized electrons in elemental gaseous states can also cause the production and emission of photons of light by undergoing transitions to lower energy states. Atomic absorption and emission spectra are unique for each element, with characteristic fixed frequencies for any transition, whether absorption to a higher energy or emission back to the lower energy. The transfer of energy causing absorption spectra is quantized among discrete orbital states.
Atomic spectra explained
Isaac Newton, after discovering the Calculus, used its principles to demonstrate the inverse square law for gravity and eventually formulated the first three fundamental laws of Physics. This led to the dramatic description of the nature of planets orbiting the sun, where at any position their centrifugal force equals the oppositely directed gravitational force, and potential and kinetic energy interchange throughout the orbit as planet velocity continuously varies in rhythmic fashion, and as the orbital radii increase for the various planets, their velocities inversely decrease. Potential energy increases to become less negative at larger radius, and kinetic energy decreases due to slower velocity. These principles also apply for electrons in atoms and molecules.
From the 3rd Law, stable orbiting bodies experience equal and opposite centripetal and centrifugal forces where mv2/r = F. For gravitational attraction, F = GmM/r2. Rearranging produces GmM/r = mv2. Since gravity is only attractive, the left side term is the negative of the integral of GmM/r2 with respect to r which represents the potential energy magnitude, and the right-side term is twice the formula for the kinetic energy of the orbiting body m. In other words, the kinetic energy of a stable orbiting body subject to Newton's laws of motion always equals half of the potential energy magnitude between the bodies. This has been referred to as the Virial theorem which has been applied to explain the behavior of physical systems [1].
Niels Bohr applied Newton's laws in quantized orbitals of the electron in the hydrogen atom, to obtain relations that compute the average radii, velocities, and energies of quantum states in the atom. Erwin Schrodinger using wave function quantum mechanics described the more detailed nature of these energy states and obtained formula for the most probable values of these parameters that are in agreement with the Bohr formula of average values. In the case of atomic energy states, the responsible force is electrical between opposite charges given by F = kqq/r2. Since electrical and gravitational forces do not dissipate to zero even at large distances, following the inverse square law, a question to consider is how large can an atomic orbital be in a stable hydrogen atom?
Discrete lines for the spectral absorption and emission of light by elemental hydrogen [2] were explained accurately by Bohr [3,4], Formula for the most probable energy and radius for an electron in all possible energy states were later obtained by Schrodinger [4] who mathematically derived the shapes of orbitals using wave functions to compute probability densities relative to radial position for the electron in its allowed energy states. Both treatments produced the same formula for most probable orbital radius and energy as a function of the principal quantum number n.
Here these formula are used to compute the most probable radius and total energy for various hydrogen orbitals to compare with the classical average energies that should exist if the potential and kinetic energies are consistent with Newton's laws, where centrifugal force mv2/r must constantly counter the centripetal force of electrical attraction. The integral of the electrical force, F = kqq/r2, along a radius produces the potential energy formula that is half the kinetic energy of the electron. Namely, ∫kqqdr/r2 = -kqq/r, and since kqq/r2 = mv2/r, then at any particular instantaneous value of r,
kqq/r = mv2 (1)
which is half the kinetic energy of the particle given by KE = ½mv2.
Orbital energies are computed in these ways and are in agreement for all orbital quantum numbers tested. The magnitude of light energy required to "ionize" the hydrogen atom from its ground state was first estimated by Johannes Rydberg (1890). The accurate Rydberg constant RH value can be computed theoretically and measured experimentally and equals the ground state orbital most probable energy. However, the difference between this value and the highest known photon energy absorbed by elemental hydrogen in vacuum is proposed here to determine the orbital quantum number to which the maximally energized electron could theoretically reside before relaxation back into lower energy states. The significance of these findings is discussed.
Methods
Current highly accurate values for constants involved in computing orbital properties for the hydrogen atom electron were used from referenced sources. For protium, 1H1, the 2p orbital electron principal quantum number is n = 2. The most probable (Schrodinger) radius of this orbital is identical to the average radius (Bohr) for n = 2 where
r = n2h2εo/πmq2 (2)
Which equals 21.16 × 10-11 m. From Equation 1, the electron average velocity is v = 1.09 × 106 m/s (0.36% of light speed), kinetic energy KE = 5.41 × 10-19 J, and average potential energy PE = -10.91 × 10-19 J with total energy Etotal = -5.5 × 10-19 J.
For the 1s orbital electron n = 1, rn = 5.29 × 10-11 m, v = 2.19 × 106 m/s (0.73% of light speed), KE = 21.84 × 10-19 J, PE = -kq2/r = -43.66 × 10-19 J, and total energy Etotal = -21.83 × 10-19 J. As expected, this value is similar to the measured value of what is considered the experimental value for the Rydberg constant RH (= -21.79 × 10-19 J) for the assumed ionization of hydrogen, but is essentially identical to the RH value calculated from the ground state Bohr or Schrodinger energy for n = 1:
Etotal = -mq4/(8εo2h2)(1/n2) = -21.82 × 10-19 J (3)
Using current accepted values of high accuracy for the electron mass m, the permittivity of free space in vacuum εo, and Planck's constant h. here,
Etotal = (9.108 × 10-31 kg) (1.602 × 10-19 C)4/[8(6.625 × 10-34 J-s)2 (8.854 × 10-12 C2/Nm2)2]
The emission line wavelengths for elemental hydrogen gas were measured with homemade spectroscopes.
Results and Discussion
Calculated and observed orbital energy agreement
To examine the accuracy of the Bohr and Schrodinger formula for the properties of the energy states of hydrogen, the theoretical energy changes for electron transitions are computed and compared to the energy of observed spectra for hydrogen. The formula for the energy and radius of the possible orbitals of the hydrogen atom as a function of the principle quantum number n were derived without use of experimentally determined quantities and thus represent theoretic ad initio descriptions. The accuracy of this formula is assessed by comparing computed energies with what is actually observed by experiment.
For the 1s 2p to transition of the hydrogen electron, the change in potential energy and the change in kinetic energy calculated from the Newtonian based Bohr average energy, which is the Schrodinger most probable energy, formula are ∆PE = 32.75 × 10-19 J and ∆KE = -16.43 × 10-19 J with a total energy change ∆Etotal = 16.32 × 10-19 J. From E = hc/λ the wavelength of light absorbed for this transition is λ = 121.8 nm. The actual observed wavelength reported for this transition, from 16.4 × 10-19 J [5], is typically reported [3] at 121 ± 40 nm. An additional source [4] for the 1s to 2p transition is 10 ev = 16 × 10-19 J which is 124 nm light. This demonstrates how accurately Newton's laws predict the characteristics of the hydrogen orbitals.
Summary of orbital characteristics
Table 1 summarizes calculations of the radii, velocities, kinetic and potential energies, and total energies for the hydrogen electron as a function of principal quantum number n. Notice that the computed values for kinetic energy are identical to half of the negative of the computed values for potential energy for each and all orbitals, consistent with the derived formula from Newton's 3rd law of motion above (Equation 1).
Average values for the kinetic energy, potential energy, total energy, and velocity for the hydrogen electron as a function of n are computed from the average radii determined by Bohr by using Newton's third law of motion with the constantly equal magnitudes of centrifugal and electrical forces (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7). These magnitudes are shown in Table 1 for values of n from 1 to 30. The sum of the KE and PE are comparable to that computed from the quantum mechanical formula for Etotal for all values of n. Magnitudes of each are shown in graphical form as a function of n for clarity.
These data emphasize the validity of Newton's Fundamental Laws of Physics that apply even for subatomic particles existing in quantized energy states. Wave mechanical treatments provide more detailed, landmark information fundamental to particle dynamics in atoms, namely that velocity, radius, KE, and PE are dynamic quantities that oscillate rhythmically for the electron as it propagates in wavelike patterns in cloud-like shaped patterns. It is necessary however to also demonstrate that this information does not detract from the accuracy of Newtonian mechanics for average values of physical quantities of submicroscopic particles. The total energy required to remove the electron from the atom always equals the kinetic energy that the electron has in any particular orbital. Further, at the ground state velocity, the electron would encircle the hydrogen atom about 2 million billion times per second, or about 2 trillion times each millisecond. In this way, matter when touched appears to be a motionless solid even though matter is made of a dynamic system of rapidly oscillating subatomic particles.
Theoretic ionization of ground state hydrogen
For the ionization of ground state hydrogen to form an "ejected" electron, we write from Newton conservation of total energy: Etotal = Einitial = Efinal since the photon of energy E = hf is completely absorbed,
PEinitial + KEinitial + hf = PEfinal + KEfinal (4)
It is typically assumed that the Rydberg constant reflects the total ionization and release of the ground state electron from the atom so that the final PE and KE are zero (where the principal quantum number approaches infinity). Using the accepted value for the Rydberg constant obtained from emission spectra for hydrogen in vacuum, Equation 4 becomes:
-43.66 × 10-19 J + 21.84 × 10-19 J + 21.79 × 10-19 J = PEfinal + KEfinal
Etotal = PEfinal + KEfinal = -0.03 × 10-19 (~ 0) J
Which is the total energy for the energized electron. Substituting PEfinal = -2KEfinal produces
Etotal = -0.06 × 10-19 (PE) + 0.03 × 10-19 (KE) = 0.03 × 10-19 J
With this kinetic energy, the velocity of the electron would be v = 8.11 × 104 m/s, 0.027% of light speed.
The difference between the energy of the ground state and the experimentally observed value of the Rydberg constant determined from emission spectra of elemental hydrogen in vacuum is often attributed to theoretical error. However, it is possible that this difference has significance. For example, a value of Etotal of zero is usually assumed for the electron after the atom is completely ionized from its ground state theoretic energy level. This however does not mean that kinetic energy and potential energy are themselves actually zero. Rather, the sum of the two approaches, but cannot equal, zero. Electric fields do not dissipate to zero even at large distances. Since infinity is not reachable, it is possible that a high energy orbital (with minimal potential energy) is technically achieved by the absorption which leaves a residual energy for the electron. Indeed, the energized electron falls back again to emit light of the same frequency and energy as the absorbed light in the first place.
If accurate, a value of -0.03 × 10-19 J is the total of kinetic energy plus potential energy for the nearly ejected electron, and this absolute value is the magnitude of the electron kinetic energy. The residual energy that the electron has after maximum light absorption transition energy is absorbed above the ground state may be used to compute an orbital quantum number n that would be created after this maximal absorption from the ground state atom, where n2 = -mq4/(Etotal8εo2h2). Using the accepted value for RH to determine the residual energy of -0.03 × 10-19 J above, this corresponds to an orbital quantum number of n ~ 27. Although it is typically assumed the absorption of this amount of Rydberg light energy, which leads to ionization of the atom, is essentially where quantum number n approaches infinity, notice that the actual value of n is finite. This is consistent with electrostatics, where no separation distance between opposite charges in vacuum would produce an electrical attraction force of zero. As discussed below, physical considerations suggest it is not likely that a single photon absorption event achieves this level of energization.
The actual magnitudes of any high energy orbital are not zero since zero energy is not attainable for the particle, so the final energy after ionization must remain a combination of PE and KE. If the total energy were actually zero, then the PE would need to be zero with the electron at infinity, and the KE would be zero, which would require the electron to be completely stationary, both of which are not possible.
Electric forces do not disappear to zero even at great distances. It is thus not surprising that the highest energy photons emitted in hydrogen gas spectra is not the same as the energy the electron has in the ground state. An orbital principal quantum number that is very large is still not infinity, and electrons cannot be made to have zero potential (or kinetic) energy. So light absorption energizes the ground state electron to a high energy, almost as high as zero, state. The difference between the energy absorbed and the ground state orbital energy is here referred to as the residual energy that the energized electron would contain after the maximum energy absorption event.
The value to 5 digit precision for the observed Rydberg constant is regarded as RH = 21.799 × 10-19 J [2] 91.17 nm light) for the 1s to"∞" transition H → H+ + e-. Here the potential energy of the electron in the ground state 1s orbital is computed from Newton as PEinitial = -43.66 × 10-19 J which changes to an assumed zero final potential energy PEfinal = ~ 0, and the initial kinetic energy KEinitial from Newton is 21.84 × 10-19 J which becomes assumed zero kinetic energy KEfinal = ~ 0. The absorbed light wavelength computed from E = hc/λ for the Newton description is λ = 91.0 nm, and from the Rydberg constant, λ = 91.17 nm (21.799 × 10-19 J).
Over many years of experimentation, students and staff at Palomar College obtained values for the Rydberg constant that are comparable to those published. RH values were computed separately for three electron transitions observed for emission lines from hydrogen vacuum tubes. For the hydrogen spectrum, the transition from n = 3 to n = 2 produced an RH value averaging 108 × 103 cm-1, the 4 to 2 transition averaged 111 × 103 cm-1, and the 5 to 2 transition is at 109 × 103 cm-1, with an average RH value of 109 × 103 cm-1 and a photon energy of 21.7 × 10-19 J, in good agreement with the average value used in the analysis here.
Experimentally observed hydrogen emission lines
The inability to find emission lines from very high quantum number orbitals is exemplified as shown in Figure 8, a plot of the recognized wavelengths of the first 26 emission lines for hydrogen in rank order from lowest wavelength (highest energy) to longest wavelength (lowest energy). The first 8 represent transitions from higher orbitals to the ground state n = 1 orbital, where the transition from n = 2 to n = 1 is the known minimum energy to this 1s orbital from the 2p orbital, with a wavelength of 122 nm. The next set of 8 lines represent emissions for transitions to the n = 2 energy state, ending with the lowest energy and longest wavelength for the 3s to 2p transition of 656.7 nm. The next 5 are transitions to the n = 3 level with the longest wavelength for the 4 to 3 transition of 1876 nm, and the next 3 transitions to n = 4, and the last two to n = 5. Allowed transitions only occur between orbitals of differing n and differing magnetic quantum number l (both ∆n and ∆1) The 15th and 16th values may represent transitions to the 2p orbital rather than the 2s, and the progressively increasing wavelengths in the n = 3, 4, and 5 sections likely reflect transitions among p, d and f sublevels as well. The energy differences between higher orbitals with quantum number greater than 5 are not sufficiently different to produce lines that are distinct and significantly intense.
There are 26 recognized emission lines for elemental hydrogen that are of significant intensity when hydrogen vacuum tube emissions are measured with spectroscope diffraction gratings [4]. The lowest energy line occurs at a wavelength of 7,485 nm, having a photon energy of -0.266 × 10-19 J. An example transition of this energy would be from n ~ 19 to n = 8. The highest energy line occurs at a wavelength of 92.6 nm, having photon energy of 21.46 × 10-19 J corresponding to a transition energy from n = 8 to the ground state. This does not represent complete removal of the electron so this energy is not equal to the magnitude of RH. Absorption and emission lines are not readily discernible at energies higher than this. Thus, electrons are not energized to an atomic size greater than 3.8 nm in a single absorption event. This is consistent with the fact that hydrogen does not absorb X-rays. Complete removal of the electron to "infinity" is not observed and "photoionization", the formation of the hydronium cation as a result of a single photon, appears unlikely in vacuum.
Because of the very accurate agreement between theory and experiment for the transitions between energy states, the difference between the energy of the 1s orbital electron and the highest observed emission line for the vacuum spectrum of hydrogen is considered significant and real. What this means is that the 1s electron is not ejected to "infinity" but rather to a possible high energy, near zero, orbital when the electron is energized through light absorption. And the emission line of highest energy represents an allowed transition from such a high orbital to the ground state. There is no emission line equal to the energy of the 1s orbital itself. It is also possible that transitions from higher, nearly removed, electron energy states occurs first to states of intermediate energy before finally transitioning to the ground state orbital. This is supported by the fact that the radial distance between the 1s orbital and orbitals of such high quantum number is relatively vast. A single transition from such a distant orbital of large radius to the ground state in a single step may not be favored over intermediate transitions instead that are more feasible.
This again suggests that ionization of atoms may be associated in vacuum with simply a very high energy orbital with a very small negative potential. This is consistent with electrostatics, where separated opposite charges have an attraction between them even at great distances. This means that the light absorption event occurs for an isolated atom in vacuum when the difference in energy between initial and final orbital states is sufficiently close to the resonant frequency of the absorbed light. The ionized electron is not completely actually ejected to infinity, but rather contains a residual energy with a negative potential energy causing the formation of an orbital, with albeit a very high quantum number, if the electron is not trapped by surrounding materials (even reacting with other substances technically does not eliminate the electrical force of attraction from the nucleus from which it was energized). The maximum energy level that may be occupied after light absorption is not certain, because orbitals from 8 to 27 and higher are so similar in energy. The emission band considered the highest energy may consist of very many very closely spaced emission wavelengths too little different to be resolved. So the absolute highest orbital to which the electron is energized by light absorption in a single event may be n = 8 but for multiple wavelength absorptions, this is not actually certain.
On the other hand, the accurate energy known for the ground state orbital theoretically corresponds to a photon energy of 92 nm, which is not achieved through light absorption experimentally. Indeed, this is in the X-ray region of the EM spectrum, and it is well known that hydrogen does not absorb or emit X-rays. This is consistent with the interpretation here, that the Rydberg energy, although accurate, is not able to be achieved through light absorption.
Defining ionization
Taken together, the information strongly suggests that the terms "ionization" and "ionization energy" do not actually mean complete removal of an electron from the electrical attraction of the nucleus of an atom. Rather, ionization refers to the process by which electrons are transferred and captured by another substance.
Radius trends are opposite energy trends as n increases
Graphs of the relative energies of hydrogen orbitals as a function of quantum number n are familiar and demonstrate how small energy differences between orbitals become as n increases beyond n = 4. On the other hand, not always appreciated is the fact the radii for those orbitals increases dramatically as n increases (Figure 9 and Figure 10). In other words, a large difference in distance between adjacent orbital positions is associated with very small differences in energy at large values of n.
This suggests that the physical distance the electron must span, for a transition to the next lower orbital, is relatively large compared to the miniscule energy difference between those orbitals so that absorption no longer occurs or is not significant. The radial span distance from n = 30 to the ground state is enormous, and absorption to higher levels is not considered reasonable, and to "infinity" is of course impossible.
Residual electron energy
The ionization energy for the hydrogen atom determined in hydrogen gas electrical ionization vacuum tubes is widely known to be approximately 21.799 × 10-19 J (91.17 nm light) determined from the slope of plots of the Rydberg equation for various values of n. This represents not only the Rydberg constant energy required to remove the 1s electron from the atom but also reflects the ability of the ionized ejected electron to undergo chemical reactions with environmental materials. This value however appears to be constant whether determined in vacuum or in air. In air, a typical reaction that the ejected electron would undergo to remain trapped off the original atom is:
(1/2) O2 + 2e- + 2H+ → H2O.
Although it would be thought that the value would be influenced by the tendency of outside materials to attract the electron from the hydrogen atom, the Rydberg constant is experimentally found to be similar in both vacuum and air. This suggests that whether the ejected electrons participate in a chemical reaction, or rather de-energize into internal orbitals in the atom or a neighboring hydrogen ion, this accurate single ionization must occur before either subsequent process takes place.
The residual energy of the energized electron creating an orbital with quantum number n = 27 would have a radius of 38 nm, about 729 times the ground state radius. At n = 8 the radius is 64 times the ground state radius. This large separation would allow the electron to easily react with other substances in the surroundings, while in vacuum the energized electron may occupy a high energy orbital, though its accurate quantum number must be estimated. However, the natural entropy requiring energy minimization to a lower orbital state prevents any prolonged residence time there. It must be emphasized that atomic orbitals described by Bohr and Schrodinger do not actually exist if no electron occupies them. Rather, the resonating electron which can exist in any of the described energy states causes the existence of the orbital.
Rydberg states
Spectral lines for emissions of light to the 1s orbital from orbitals of high values for n are not experimentally distinguishable. So the highest orbital involved in the transition to the 1s is not necessarily the highest orbital the electron can occupy. Indeed, energy differences between adjacent orbitals for n values greater than 6 are quite minute. So although it is not possible to detect emissions from such high energy (near zero) orbitals, these orbitals can certainly exist when occupied. The energy differences between high value orbitals are small compared to the large radial distance between them. So light absorption between very high energy states approaching zero energy would not occur. The electron cannot have truly zero energy with a proton in the vicinity. Unless the energized electron is extracted by an adjacent substance, it may occupy a high quantum number orbital. If this energized electron returns to the ground state, this would not produce a discernibly unique emission line. In this case the reactions may be summarized as:
H2(g) + 2hf(absorbed) → 2H+ + 2e- → H2(g) + hf(emitted)
Although the overall equation appears to suggest that light hf is a catalyst, it is not. The absorbed light has a velocity into the atom while the released light velocity is directed outward and is responsible for emission lines detected in spectra of the atom. Since UV light homolytic cleavage of hydrogen gas into isolated radical hydrogen atoms which follow the energy formula used here, emission lines are used for the analysis because these reflect the orbital transition back to ground state within the isolated atom.
One possibility is that the highest photon energy absorbed has to do with the fact that transition energies larger than this, which involve very much larger radial distances between states, may not be possible through light absorption. Since it is not possible to energize an electron "to infinity", it should not be surprising that this difference exists. It might not ever be possible to absorb light energy matching the ground state energy because light absorption alone might not actually ionize the atom. Indeed, the highest energy light observed in emission spectra, associated with relaxing transitions, is also the same as for the maximum absorption energy transition, consistent with this possibility. Indeed, Rydberg or high energy states of atoms are known to exist [6].
If one assumes for argument that the difference between computed ground state and experimental Rydberg energy is significant, then it is possible to compute the quantum number energy state to which an electron would be energized if this were the residual energy the electron had after maximum energy light absorption. The energy difference must equal, from the Virial theory, the most probable kinetic energy of the maximally energized electron, and this corresponds, with the Bohr/Schrodinger formula, to a value of n = 27. The difference between ground state computed energy and the highest energy emission line observed corresponds to a high energy state where n = 8. Both considerations indicate that instead of the maximum light energy forming an ionized atom, the electron is energized to a high energy Rydberg state, rather than "to infinity".
Of course, if there are environmental substances or receptors present, such as surrounding positive charges in the sample, the electron could be trapped by that substance and this would be true ionization. But the above argument suggests that this state might not be achieved through light absorption alone. The possibility that it is the theoretic formula for electron properties that must be adjusted to match the experimental data is disputed by the perfect agreement between Bohr and Schrodinger expressions and experimental emission energies for transitions among energy states, other than the putative transition to "infinity". Any theoretic correction to the nature of the energy levels would affect all levels of the hydrogen atom, not simply the transitions in question. Taken together the difference between the theoretic and experimental highest possible energy transition magnitudes is not due to theoretic error but rather to the perceived view that this transition represents complete ionization to "infinity".
Instead of stating that the ionization of hydrogen is associated with the electron being removed or energized to an orbital with a quantum number approaching infinity, it should perhaps be said that the orbital to which the electron can be excited, by the maximum observed energy absorbed, has a high but finite quantum number. Estimates using currently accepted values for the Rydberg constant suggest the orbital quantum number may be as high as n = 27. The energy required to theoretically totally remove electrons from high energy orbitals (near zero energy) is plotted in Figure 8 for principal quantum numbers up to n = 30. Note the relative energy differences between n = 28 and n = 30 orbitals is miniscule compared to differences between lower quantum number orbitals. This, together with the fact that the radial distance to the n = 27 orbital is 729 times further from the nucleus than the ground state orbital, may explain why the highest energy emission line observed in vacuum hydrogen spectra is limited and is significantly less than the ground state energy. Note that the energy change between n = 27 and n = 30 is a mere 0.03% of the 1s orbital energy, while the corresponding change in radius is a huge 20% of the original 1s orbital radius.
The radial distance between the ground state to the n = 42 level is an average of 93 nm. This orbital is the first at which this distance exceeds the wavelength of the photon that would induce the transition, at λ = 91.1 nm. Although this may be an inconsequential coincidence, one possibility is that this is a physical reason why photon absorption is not detected from the ground state to orbitals this high. This is shown in Figure 11, which is a graph of the wavelength of light required to induce a transition from the ground state, and the radial distance from the ground state to that orbital, both as a function of principle quantum number n. An electron from the ground state does not transition to a radial distance greater than the wavelength of the light required for the transition. Absorption of photons only occurs if the wavelength of the photon is no larger than the radial distance between energy states. The wavelength of the photon required to induce such a transition is smaller than the radial separation distance to that orbital. All experimentally-observed transitions occur when the radial distance between energy states is less than the photon wavelength required for the resonant transition. In the photoelectric effect, shorter wavelengths than threshold for ejection add extra energy after release in a metal wire. This demonstrates how vastly different is the structure and behavior of the electron in elemental hydrogen gas from the 'sea' of delocalized electrons present in metals or the plasma on the sun that emits a broad, continuous spectrum of radiation.
Although these values for n seem large, none of course approach a transition "to infinity." The linear energy density (hf/λ) of photons for this presumed maximal transition is ~1 × 10-22 J/m. This is between the linear energy density for UV light centered at 7 × 10-26 J/m and infrared light at 2 × 10-16 J/m.
If an energized electron in a very high energy orbital returns to the ground state, this would not produce a discernibly unique emission line because the difference in energy between high energy orbitals becomes miniscule and any low probability emission becomes too faint to be observed. And in theory no emission of light could be observed that equals the energy of the ground state orbital, the magnitude of the Rydberg constant for complete elimination of the electron, because it is not possible to exist in an infinite orbital at an infinite radial distance from the proton. High energy states of atoms usually with a quantum number greater than n = 30, referred to as Rydberg states [6,7], are known. These high energy orbitals are not at an infinite distance and do not have a quantum number of infinity, but rather are large atoms with energized electron energies near, but not equal to, zero. These atoms can remain energized for several nanoseconds before returning to lower energy states and are so large that Bohr circular orbital descriptions apply, thus bridging the gap between subatomic and cosmologic structures [7].
Figure 12 is a plot of the radial distance between adjacent orbitals per light energy associated with that transition, from n = 2 to n = 27. The distance between adjacent orbitals becomes vast compared to the energy of the light required to induce that transition or to be emitted from it, so these transitions are not physically probable.
The finding, that the Rydberg energy required for complete elimination of the hydrogen electron from its ground state by resonant light absorption is accurate but not equal to the energy of the largest absorbed light energy transition observed, is not surprising. There is a comparable precedent. Cooling an ideal gas decreases its volume by a factor of 1/273 for each 1 ℃. Extrapolating this change to 0 K is the temperature at which the gas volume would equal zero and is correctly at -273.15 ℃. Since no gas can be cooled until its volume disappears, absolute zero cannot be reached by a real gas molecule through cooling. Similarly, since no electron can be ejected to infinity, the Rydberg theoretic constant needs no corrections but cannot be reached through resonant absorption of light by hydrogen.
Comparisons between atomic orbitals and planetary orbits
It is important to realize that planets in the solar system have so much in common with this subatomic energetic behavior [8]. The atomic orbitals of electrons around the nucleus of the hydrogen atom have features strikingly in common with the orbits of the planets around the sun. Although orbit energy for the planets depends on the widely differing planet masses, plots of velocity as a function of planet distance from the sun mirror those for the electron in various energy orbitals. The planet Mercury in its orbit travels at an average velocity that is ten times faster than Pluto, while the radius of the Pluto orbit is 100 times larger than that for Mercury. Likewise, the electron in the ground state orbital of hydrogen travels at an average velocity that is ten times faster than the electron in the orbital with quantum number ten, while the radius of this orbital is 100 times larger than the ground state orbital. Likewise the moon is 14.6 times further from the earth center than a GPS satellite is, and the moon orbit speed is 3.8 times slower. These features of both the macro and the subatomic realms testify to the accuracy of the inverse square law for both electric and gravitational forces, and the vast applications of the Fundamental Laws of Physics that Newton discovered.
Cosmologic significance
The analogy to the solar system is that very distant orbiting objects should exist at great distances up to the position where an adjacent solar system does not prevent its stable orbit in the other. Such an example might be Halley's comet which navigates our solar system at an aphelion distance of 5.27 x 109 km. As far as galaxies in the universe, Newton's 3rd law suggests that the entire universe of matter may be in a state of permanent rotational motion, where centrifugal forces due to lateral velocity and centripetal forces due to gravitational attraction acting at great distances, form a stable universe in a dynamic state of equilibrium. This is possible, where such a notion has not been convincingly disproven. Although the Virial theory, derived from Newton's 3rd law, has been used to estimate the mass of the universe, from M = rv2/G, and has suggested to some the existence of unknown dark energy or dark matter to explain disagreements between estimated and observed velocities of galaxies, it must be emphasized that only lateral velocity components produce the centrifugal radial force, so receding velocities should not be so simply applied. The notion that the entire system of galaxies is now and has long been expanding or receding in space on a net basis is uncertain. It is not possible to stop a galaxy in space to determine that it is radial motion and a Doppler shift alone that explains observed wavelengths of emitted light from distant stars.
The recently developed map by NASA from the Planck Project of the cosmic background radiation of the entire skyline indicates that the universe of galaxies forms an ordered shape. Individual galaxies have ordered structures, many being named for their shapes, such as the dramatic Sombrero galaxy. And our own solar system lies on a planar ecliptic. Taken together, this refutes the notion that all matter was at one time together that exploded and expanded. Rather, it appears that the entire universe undergoes rotation to prevent galaxies from collapsing into each other due to gravity, fully consistent with Newton's laws. Much like electrons continuously propagating around central nuclei, and planets around the sun, with velocity and energy changing continuously, the entire universe of matter may also be in such a dynamic equilibrium steady-state. Just as there is no atomic electron orbital at infinity, there is no galaxy that resides at infinity either. The universe of space is infinite, and the universe of matter resides within that space. Gravity from mass, like electrical force from charge, does not become eliminated at any great distance (Additional Information).
Acknowledgments
Isaac Newton is appreciated for spending an entire lifetime investigating the nature of the universe and concluding it is like a vast Created wound clock. Much gratitude goes to faculty at the University of California, San Diego, La Jolla, CA who taught us in the 1960's when the University first formed. Thanks go to Professor Ed Kentner, Palomar College for his expert chemistry teaching for so many years. And thanks go to my Pearl Harbor dad for encouraging me to "stay in school."
Disclaimer
Viewpoints presented here do not necessarily reflect the views held by Palomar College or its staff. No conflicts of interest are involved in this work.

