
International Journal of Nanoparticles and Nanotechnology
(ISSN: 2631-5084)
Volume 6, Issue 1
Original Article
DOI: 10.35840/2631-5084/5537
Functional Cotton Fabric: Enhancement in Flame Retardancy and Thermal Stability
Ahmed Abed1,2,3*, Nabil Bouazizi1,4*, Stéphane Giraud1, Ahmida El Achari1, Christine Campagne1, Julien Vieillard4 and Franck Le Derf4
Table of Content
Figures

Figure 1: Optical microscopy of CO (a-b) and CO-PVDF-TiO2-CT....
Optical microscopy of CO (a-b) and CO-PVDF-TiO2-CT (c-f) composites and its SEM image.

Figure 2: Photograph of colored water immersion-height.....
Photograph of colored water immersion-height during 60 min and changes in drop shape on: a) CO; b) CO-PVDF-TiO2-CT composites.
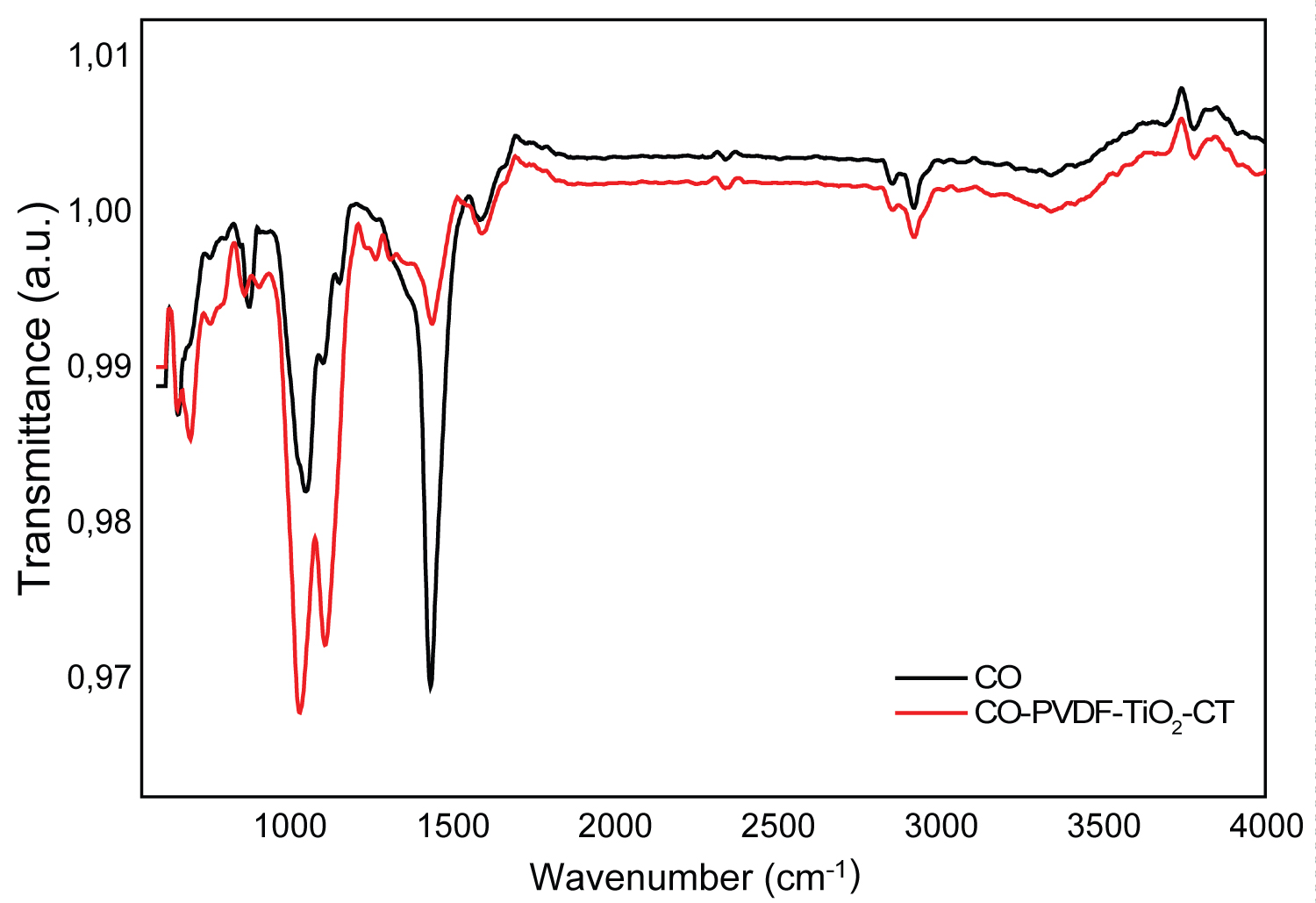
Figure 3: FTIR spectra of CO and CO-PVDF-TiO2-CT composites....
FTIR spectra of CO and CO-PVDF-TiO2-CT composites and the XRD pattern of CO.
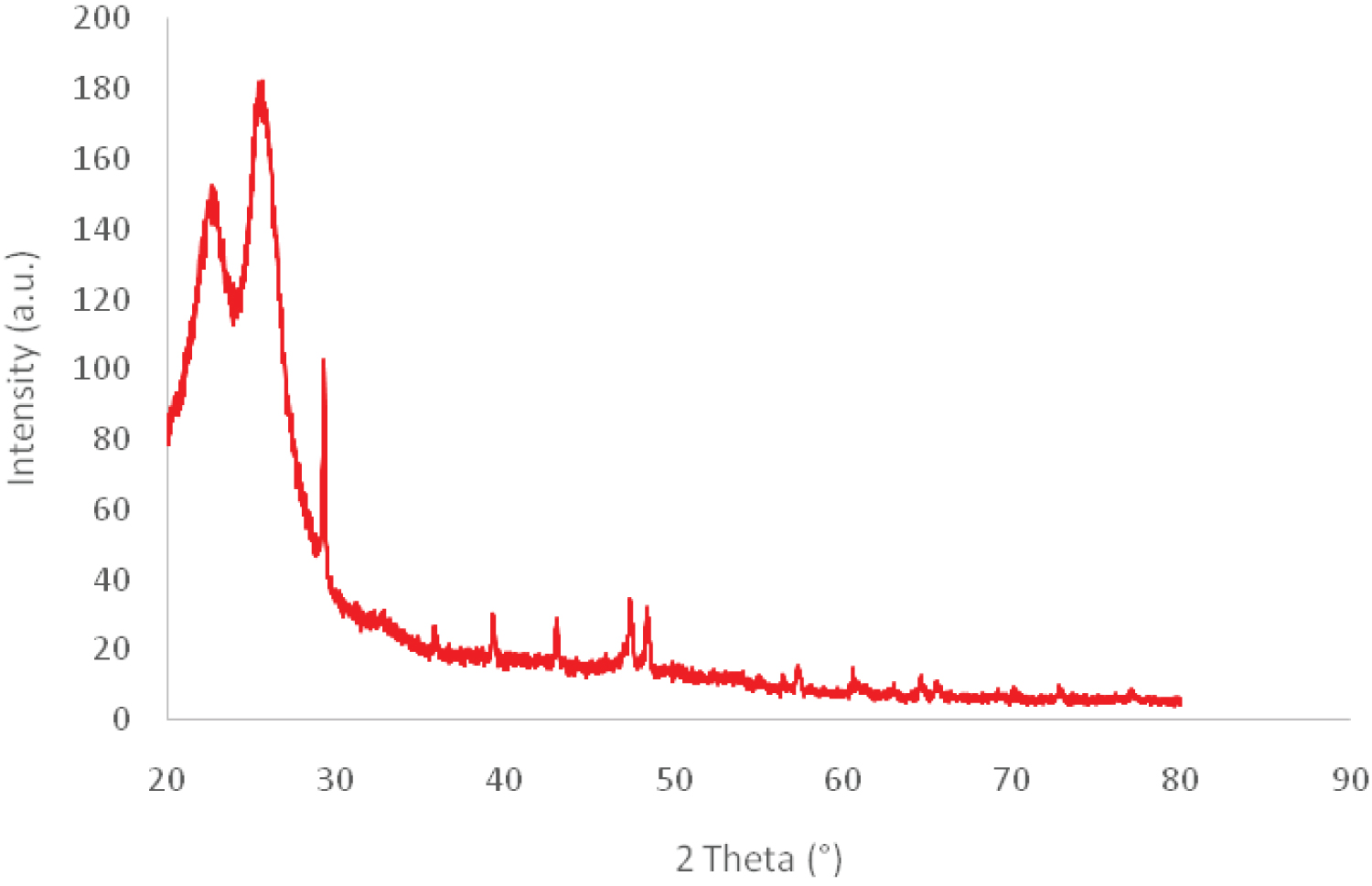
Figure 4: TGA patterns of original cotton fabric....
TGA patterns of original cotton fabric (a) and CO-PVDF-TiO2-CT composites.

Figure 5: Flame retardancy tests on original CO....
Flame retardancy tests on original CO: a) Before; b) After ignition, and on the CO-PVDF-TiO2-CT composites; c) Before and; d) After ignition.
Schemes
Scheme 1: Postulated ternary system as composite for flame....
Postulated ternary system as composite for flame retardancy application.

Scheme 2: The synthesis and functionalization steps.....
The synthesis and functionalization steps of biobased cotton.
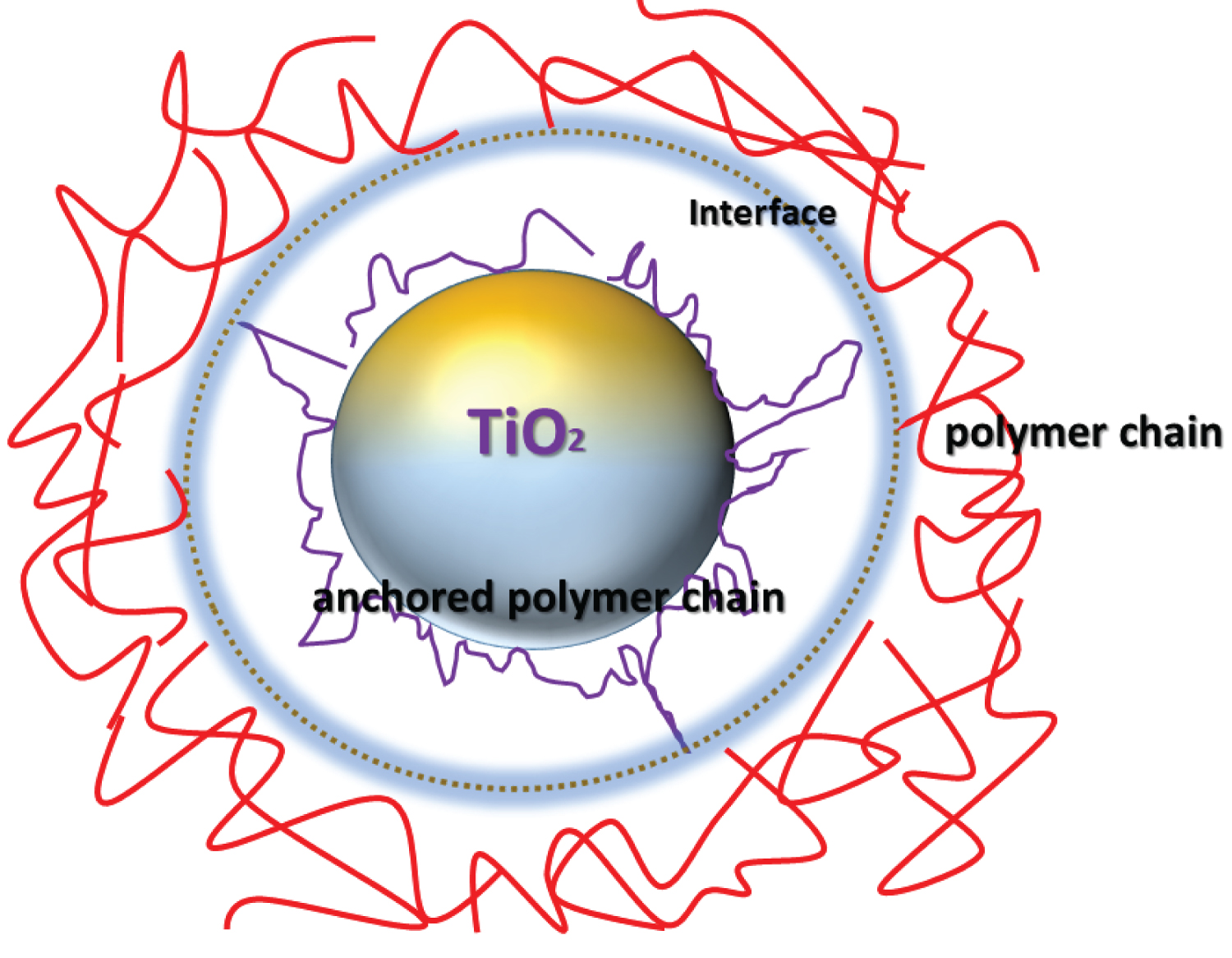
Scheme 3: Proposed adsorption of polymer chains on the surface....
Proposed adsorption of polymer chains on the surface of the metal oxide.
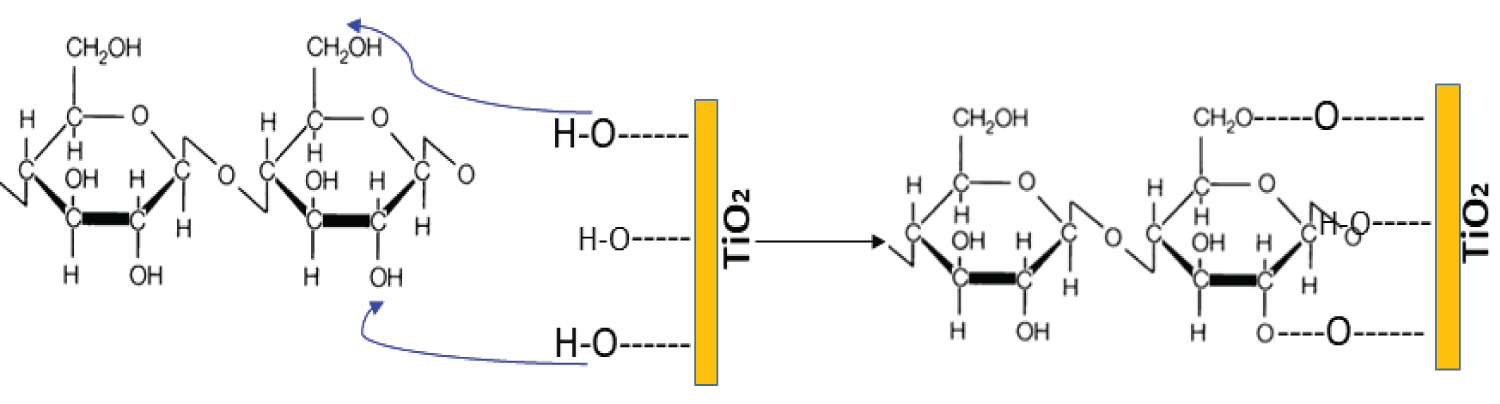
Scheme 4: Possible intervention of hydroxyl groups present....
Possible intervention of hydroxyl groups present on the surface of TiO2 particles with cotton structure.
Tables
Table 1: Evolution in time of the contact angle for CO and modified counterparts.
Table 2: TGA data of CO and its modified counterparts.
Table 3: Main features in flame retardancy test of cotton fabric and its modified counterparts.
Table 4: Main features in flame retardancy test of CO and its modified counterparts.
References
- Kemmlein S, Hahn O, Jann O (2003) Emissions of organophosphate and brominated flame retardants from selected consumer products and building materials. Atmos Environ 37: 5485-5493.
- Ling HL, Yang JW, Xiang L, Li FY, Luo ML, et al. (2012) The synthesis and application of a high performance amino resin nanocomposite as leather flame retardant. J Soc Leath Technol Chem 96: 5.
- Jiang YP, Li JX, Li B, Liu HY, Li ZJ, et al. (2015) Study on a novel multifunctional nanocomposite as flame retardant of leather. Polym Degrad Stabil 115: 110-116.
- Wegene JD, Thanikaivelan P (2018) Synergy of organic nanoclay and inorganic phosphates for fire retardant leather applications. J Am Leather Chem Assoc 113: 371-379.
- Xu WL, Li JX, Liu FM, Jiang YP, Li ZJ, et al. (2017) Study on the thermal decomposition kinetics and flammability performance of a flame-retardant leather. J Therm Anal Calorim 128: 1107-1116.
- Van der Veen I, de Boer J (2012) Phosphorus flame retardants: Properties, production, environmental occurrence, toxicity and analysis. Chemosphere 88: 1119-1153.
- Abed A, Bouazizi N, Giraud A, Achari A El, Campagne C, et al. (2019) Preparation of a novel composite based polyester nonwovens with high mechanical resistance and wash fastness properties. Colloids and Surfaces A: Physicochemical and Engineering Aspects577: 604-612.
- Azimi R, Roghani-Mamaqani H, Gholipour-Mahmoudalilou M (2017) Grafting poly (amidoamine) dendrimer-modified silica nanoparticles to graphene oxide for preparation of a composite and curing agent for epoxy resin. Polymer 126: 152-161.
- Jiang SD, Tang G, Chen J, Huang ZQ, Hu Y (2018) Biobased polyelectrolyte multilayer-coated hollow mesoporous silica as a green flame retardant for epoxy resin. Hazard Mater J 342: 689-697.
- Chen L, Wang YZ (2010) A review on flame retardant technology in China. Part I: development of flame retardants. Polym Adv Technol 21: 1-26.
- Veen I, Boer J (2012) Phosphorus flame retardants: Properties, production, environmental occurrence, toxicity and analysis. Chemosphere 88: 1119-1153.
- Yang G, Wu WH, Wang YH, Jiao YH, Lu LY, et al. (2019) Synthesis of a novel phosphazene-based flame retardant with active amine groups and its application in reducing the fire hazard of Epoxy Resin. J Hazard Mater 366: 78-87.
- Wang R, Zhuo D, Weng Z, Wu L, Cheng X, et al. (2015) A novel nanosilica/graphene oxide hybrid and its flame retarding epoxy resin with simultaneously improved mechanical, thermal conductivity, and dielectric properties. J Mater Chem 3: 9826-9836.
- Rakotomalala M, Wagner S, Doring M (2010) Recent developments in halogen free flame retardants for epoxy resins for electrical and electronic applications. Materials 3: 4300-4327.
- Abed A, Bouazizi N, Giraud S, Achari A El, Campagne C, et al. (2020) Polyester-supported chitosan-poly(vinylidene fluoride)-inorganic-oxide-nanoparticles composites with improved flame retardancy and thermal stability. Chinese Journal of Polymer Science 38: 84-91.
- Costa FR, Wagenknecht U, Heinrich G (2007) LDPE/Mg-Al layered double hydroxide nanocomposite: Thermal and flammability properties. G Polym Degrad Stab 92: 1813-1823.
- Plentz RS, Miotto M, Schneider EE, Forte MMC, Mauler RS, et al. (2006) Effect of a macromolecular coupling agent on the properties of aluminum hydroxide/PP composites. J Appl Polym Sci 101: 1799-1805.
- Xie Y, Hill CAS, Xiao Z, Militz H, Mai C (2010) Silane coupling agents used for natural fiber/polymer composites: A review. Compos Part A: Appl Sci Manuf 41: 806-819.
- Bouazizi N, Ajala F, Bettaibi A, Khelil M, Benghnia A, et al. (2016) Metal-organo-zinc oxide materials: Investigation on the structural, optical and electrical properties. J Alloys Compd 656: 146-153.
- Bouazizi N, Khelil M, Ajala F, Boudharaa T, Benghnia A, et al. (2016) Molybdenum-loaded 1,5-diaminonaphthalene/ZnO materials with improved electrical properties and affinity towards hydrogen at ambient conditions. Int J Hydro Energy 41: 11232-11241.
- Wei YX, Deng C, Zhao ZY, Wang YZ (2018) A novel organic-inorganic hybrid SiO2@DPP for the fire retardance of polycarbonate. Polym Degrad Stab 154: 177-185.
- Wang D, Song L, Zhou K, Yu X, Hu Y, et al. (2015) Anomalous nano-barrier effects of ultrathin molybdenum disulfide nanosheets for improving the flame retardance of polymer nanocomposites. J Mater Chem A 3: 14307-14317.
- Morshed MN, Shen X, Deb H, Azad SA, Zhang X, et al. (2018) Sonochemical fabrication of nanocryatalline titanium dioxide (TiO2) in cotton fiber for durable ultraviolet resistance. J Nat Fibers 17: 41-54.
- Uddin Khan MA, Mariko A, Orlando JR (2017) Hybrid films of chitosan, cellulose nanofibrils and boric acid: Flame retardancy, optical and thermo-mechanical properties. Carbohydrate Polymers 177: 13-21.
- Zhang S, Liu X, Jin X, Li H, Sun J, et al. (2018) The novel application of chitosan: Effects of cross-linked chitosan on the fire performance of thermoplastic polyurethane. Carbohydrate Polymers 189: 313-321.
- LV Z, Yi-Ting HU, Jin-Ping G, Ren-ChengT, Guo-Qiang C (2019) Preparation of a flame retardant, antibacterial, and colored silk fabric with chitosan and vitamin B2 sodium phosphate by electrostatic layer by layer assembly. Materials Letters 241: 136-139.
- Mohamed H, Mohamed N, Yasser A, Makhlouf G, Abdelkhalik A (2016) Synergistic effect of chitosan-based flame retardant and modified clay on the flammability properties of LLDPE. Polymer Degradation and Stability 133: 8-15.
- Marcus L, Anas A AO, Sarah CR, Grunlan JC (2015) Water-based chitosan/melamine polyphosphate multilayer nanocoating that extinguishes fire on polyester-cotton fabric. Carbohydrate polymers 130: 227-232.
- Nabil B, Ahmed A, Stephane G, El Achari A, Campagne C, et al. (2020) Development of new composite fibers with excellent UV radiation protection. Physica E: Low-dimensional Systems and Nanostructures 118: 113905.
- Gajanan DP, Aravind HP, Sushilkumar AJ, Patila CR, Patil SP (2019) A new method to prepare superhydrophobic cotton fabrics by post-coating surface modification of ZnO nanoparticles. Materials Letters 255: 126562.
- Bouazizi N, Ahmida El A, Campagne C, Vieillard J, Abdelkrim A (2018) Polyfunctional cotton fabrics with catalytic activity and antibacterial capacity. Chemical Engineering Journal 351: 328-339.
- Takke V, Behary N, Perwuelz A, Campagne C (2009) Studies on the atmospheric air-plasma treatment of PET (polyethylene terephtalate) woven fabrics: Effect of process parameters and of aging. J Appl Polym Sci 114: 348-357.
- Ahmed HB, Emam HE (2016) Layer by layer assembly of nanosilver for high performance cotton fabrics. Fiber Polym 17: 418-426.
- Lansk B (1994) Thermo-oxidation of lactam-based polyamides with carboxylic end-groups. Decomposition of 6-hydroperoxy-6-hexanelactam in the presence of carboxylic acids. Eur Polym J 30: 197-204.
- Hagler AT, Lapiccirella A (1976) Spatial electron distribution and population analysis of amides, carboxylic acid, and peptides, and their relation to empirical potential functions. Biopolymers 15: 1167-1200.
- AS Fomenko, TM Abramova, EP Dareva, AA Beloded, JG Furman (1968) Irradiation-chemical conversion of polyamide 6 and some aliphatic amides. Vysokomol Soedin A10: 507-514.
- Han Z, Dong L, Li Y, Zhao H (2007) A comparative study on the synergistic effect of expandable graphite with APP and IFR in polyethylene. J Fire Sci 25: 79-91.
Author Details
Ahmed Abed1,2,3*, Nabil Bouazizi1,4*, Stéphane Giraud1, Ahmida El Achari1, Christine Campagne1, Julien Vieillard4 and Franck Le Derf4
1ENSAIT, GEMTEX - Laboratoire de Génie et Matériaux Textiles, Lille, France
2Laboratory LPMC, Faculty of Science El Jadida, Chouaib Doukkali University, El Jadida, Morocco
3Laboratory REMTEX, ESITH, Route d'El Jadida, Casablanca, Morocco
4Normandie Univ., UNIROUEN, INSA Rouen, CNRS, COBRA (UMR 6014), Evreux, France
Corresponding author
Abed Ahmed, ENSAIT, GEMTEX - Laboratoire de Genie et Materiaux Textiles, F-59000, Lille, France; Laboratory LPMC, Faculty of Science El Jadida, Chouaib Doukkali University, El Jadida, Morocco; Laboratory REMTEX, ESITH, Route d'El Jadida, km 8, BP 7731, Oulfa, Casablanca, Morocco;
Bouazizi Nabil, ENSAIT, GEMTEX - Laboratoire de Genie et Materiaux Textiles, F-59000, Lille, France; Normandie Univ., UNIROUEN, INSA Rouen, CNRS, COBRA (UMR 6014), 55 rue Saint Germain, 27000, Evreux, France
Accepted: November 07, 2020 | Published Online: November 09, 2020
Citation: Abed A, Bouazizi N, Giraud S, El Achari A, Campagne C, et al. (2020) Functional Cotton Fabric: Enhancement in Flame Retardancy and Thermal Stability. Int J Nanoparticles Nanotech 6:037
Copyright: © 2020 Abed A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The mains objective of this work was the surface modification of cotton fabric (CO) by chemical grafting of chitosan (CT), and immobilization of titanium dioxide (TiO2) via polyvinylidene fluoride (PVDF) as binder. The resulting composites CO-TiO2-PVDF-CT were characterized and used as a flame retardant material. The thermal degradation and surface modification properties were investigated. It was found that CO-TiO2-PVDF-CT are highly hydrophobic composite with good diffusion resistance. PVDF and CT appears to be responsible for the improvement of both flame retardancy and the thermal degradation. The prepared functional composite enables it a promising candidate for aerospace and transportation applications.
Keywords
Flame retardant, Cotton fabric, Thermal degradation, Surface functionalization
Introduction
In last decade, flame retardants (FR) were used in polymers to delay the spread of fire after ignition [1]. Several works aiming at the inorganic and organic materials with flame retardant [2-5]. Recently, phosphorus flame retardant, nitrogen, zeolites and metal oxides have received a considerable attention. Particularly, flame retardant based on metal oxide was often divided into inorganic and organic flame retardants [6], which were considered as efficient alternatives for textile flame retardants. Among these metal oxides, titanium dioxide (TiO2), as flame retardant was considered a promising flame retardancy with a low toxicity. Abed, et al. [7] reported a number of inorganic oxide compounds that investigated as FR for polyester nonwoven. The results showed that metal oxide materials were a potential layer flame retardant for textile. In order to improve the flame retardancy of inorganic materials, there are numerous strategies and techniques, for instance, by grafting of halogenated FR and phosphorus intumescent to reduce costs and improve FR performance [8,9]. Nevertheless, the halogenated compounds are highly harmful and banned in industrial applications [10]. In this regard, natural polymers FR are "greener" type of additives that produce less toxicity during the combustion [11,12]. Unfortunately, inorganic FR have poor compatibility and low flame-retardant efficiency [13,14]. Therefore, many modification methods have been used to increase the flame-retardant efficiency of inorganic FR. Recently researches have been focused on developing advanced composites based on polyester and cotton fabrics with improved FR properties. Abed, et al. have found that adding ZnO and SiO2 enhances the FR of poly (vinyl chloride) microfibers [15]. Metallic oxides were activated and modified by addition of polymers and amine derivate for enhancing stability and adsorption properties [15-19]. Due to their excellent flame retardancy, chemical and ecofriendly properties, polyvinylidene fluoride were widely used various applications such as the microfiltration and in advanced water separation and environmental remediation processes. Therefore, composites based on PVDF and inorganic oxides provided high thermal stability, FR properties, and impact resistance. This was explained by the key role of PVDF, which incorporates metallic oxide, but also by the possible crosslink with the material [20-22]. In order to ensure the surface compatibility and the solid crosslinking between the composite species, researchers were employed some natural polymers like chitosan which show many advantages over artificial materials [23]. The chitosan was considered as efficient candidate in total biodegradability, biocompatibility with plants, antimicrobial, non-toxicity and fiber formation properties. In addition, chitosan appears to be the most prospective material for many fields as wastewater treatment, pulp and paper, agriculture, and textile fields [24-28]. To the best of our knowledge, studies relating a combination of all these FR additives have not been reported. In this direction, the present work aims on the development of new composite based on titanium dioxide, polymers and biobased cotton fabric as ternary system for flame retardancy (Scheme 1). An alternative chemical strategy was employed to functionalize the cotton fabric surface to produce effective composites with improved thermal stability and flame retardancy. The morphologies, thermal stability and surface compatibility were investigated in details. At last, the effect of FR and toxicity evaluation were studied.
Materials and Methods
Chemicals
Natural chitosan (CT) as biopolymers, poly (vinylidene fluoride) (PVDF) and titanium dioxide TiO2 were purchased from Sigma-Aldrich. Acetone and N, N-dimethylformamide (DMF), ethanol, sodium triphosphate (PPT), distilled water, ammonia hydroxide (NH4OH, NH3 basis), sodium hydroxide (NaOH) were purchased and used as obtained. Biobased cotton fiber were purchased from Subrenat (Ref. UB1-FROM/ CR1.2).
Cotton fabric preparation and functionalization
Scheme 2 showed the different steps of composites preparation and functionalization. The process mainly consisted of the following steps:
(i) The cotton fabrics were cleaned to remove oil impurity, according our previously published works [29].
(ii) PVDF and TiO2 were mixed in acetone and DMF (45:5 v/v) as solvent, and then stirred during 2 h at 50 °C upon the completely dissociation of TiO2. The DMF solvent was used to maintain the homogeneity of PVDF during the coating process.
(iii) CO fabrics were incubated in the above mixture solution for 2 hours under mechanical stirring. After that, the coated cotton materials were taken out from the solution and dried at 50 °C during 24 h to clean and remove residual solvent. The resulting composites denoted CO-PVDF-TiO2.
(iv) Finally, the CO-PVDF-TiO2 composites prone to the chemical immobilization of chitosan (CT) by padding via an aqueous solution of 3 g/L chitosan biopolymer in the presence of sodium triphosphate (PPT). The final product CO-PVDF-TiO2-CT were dried at 110 °C during 3 min and cured at 170 °C for 30 second using a hot air dryer (Werner Mathis AG, Switzerland).
Characterizations
The CO-PVDF-TiO2-CT and its intermediate materials were investigated via physical and chemical characterization. Fourier transform IR spectroscopy was carried out using a Tensor 27 (Bruker) spectrometer with a ZnSe ATR crystal. The samples were analyzed by FTIR directly without any further preparation, and background spectra were recorded on air. For field emission scanning electron microscopy (SEM) using a JEOL JEM-ARM200F HR setting with a field emission gun and probe aberration corrector, materials were previously metallized by a gold layer at 18 mA for 360s with a Biorad E5200 device. Thermal analysis was measured by thermo gravimetric analysis (TG-DTA, A6300R). The water contact angle measurements were carried out by using Digidrop device from GBX. This instrument uses a goniometric method to calculate the contact angle between water and a solid surface.
Results and Discussions
Morphological and structural properties
A first overview on the surface morphology of functionalized composites was obtained by SEM analysis, and the results are shown in Figure 1. The original cotton fabric exhibits a non-uniform network of overlapped fibers and multiple connected pores with a smooth surface. The sizes of the fiber were changed between 21 μm and 15 μm and a visible void were observed in the entire fabric surface. After the modification steps, the fiber of the composites appears to be linked the one with the other. This behavior can explained by the successful coating steps through the addition of PVDF, CT and TiO2. As seen, the microstructure presents a continuous film structure with a thickness layer about few microns, forming a solid contracted surface. Herein, the key role of the functionalization strategy is the coherence and the compatibility of the composite compounds. This was supported by the totally covered fiber by the PVDF/TiO2/CT layers. In this regard, it was supposed that interfacial interactions between CO, PVDF, TiO2 and CT are involved. From optical and SEM images, it is clear that uniform distribution with high particles density were immobilized at the fabric surface. Consequently, this was resulted in a much smoother surface structure and the formation of bulkier clusters of much smaller particles. Consequently, these changes can be associated to the strong interaction between CO, PVDF, TiO2 and CT, which promotes structure morphology with tailored surface.
Another confirmation was obtained from optical microscopy, indicating the good preponderance of uniform aggregates film at the fiber surface. Therefore, it appeared that both PDFV-MOx and CT successfully covered the CO fiber, in agreement with FTIR analysis, contact angle and TGA analysis.
Hydrophobic properties
In order to evaluate our developed CO-PVDF-TiO2-CT composites for environmental application, wettability and diffusion resistance are of great importance. The contact angle and capillarity of the fabric surface were investigated and the results are reported in Figure 2. Results demonstrated that water contact angle of original CO and CO-PVDF-TiO2-CT are 1.97° and 115°, respectively. Accordingly, these results evidence the highly hydrophobic properties of functionalized CO. This result is of great importance, as composite with good hydrophobic characters would be beneficial for the enhancement not only the fire retardancy properties, but also the thermal stability and durability. Deeper insights in the changes of the apparent contact angles with time were investigated, and the results were summarized in Table 1. From these measurements, the obtained results, it was established that PVDF/TiO2/CT are responsible for the hydrophobicity characters, as compared to the starting samples. Even, during 1 hour, the water cannot penetrate in CO-PVDF-TiO2-CT samples, due to the high hydrophobicity of PVDF and CT polymers. Explanation resides on the free and unlinked atoms like F from PVDF, Ti from TiO2, and N from CT, which increases the hydrophobicity. Interestingly, the above results confirm, again, the key of chitosan that ensure the crosslink between unlinked atoms and hydroxyl groups from chitosan. Thus, the hydrophobicity was slightly increased after CT grafting. This was confirmed by the droplet volume and the contact angle values, which were unchanged in time.
FT-IR evidences for tailoring surface
Infrared studies were carried out to ascertain not only the purity and nature of the functional groups, but also the coherence between all compounds of resulting composites. The FTIR spectra of original and functionalized CO fabric are shown in Figure 3. The structure of original cotton displayed some bands in the region 1720-650 cm-1, attributed to the stretching vibration of CH2, C=O, and aromatic C=C. The peaks obtained between 900 and 1285 cm-1 corresponded to asymmetric out-of-phase ring stretch: C1-O-C4 glucosidic bond, 1060 to 1065 cm-1 C-O stretching and 1285 cm-1 due to C-H deformation stretch [30]. This was in agreement with the XRD (Figure 3) which revealed the characteristic peaks of cellulose at 2 Θ =22.8° and 29.6° [31].
The broad peaks appeared at around 3710 cm-1 and 1442 cm-1 were attributed to the stretching vibrations of OH groups, corresponding to adsorbed water molecules. Afterward, PVDF fixation, the bands obtained at 869 cm-1 corresponds to the vibrations of υ CF2 for amorphous PVDF. In other hand, the impregnation of TiO2 nanocharges was confirmed by the band registered at 742 cm-1, indicating the presence of stretching vibration of Ti-O. Chitosan grafting induced a shift of the υ CF2 peaks from 889 cm-1 to 909 cm-1 because of the restriction on the vibration by the interaction occurred between TiO2, CT and CF2 groups. The peak obtained at 1597 cm-1 was assigned to the NH2 groups bend scissoring, confirming that chitosan were successfully coated the modified cotton fabrics [32,33].
Thermal analysis
The TGA curves of all samples are shown in Figure 4. It was found that two different stages were appeared between 380 °C and 400 °C, indicating the decomposition and degradation of CO-PVDF-TiO2-CT composites. The first weight loss at 423 °C for CO-TiO2-PVDF-CT and towards 435 °C for CO-TiO2-PVDF composites were attributed to the formation of residues or oxidation of volatiles species [29,31]. By comparing the analysis between samples, its clear CO-PVDF-TiO2-CT registered a favored decomposition, due to the disappeared free oxygen. These results can be explained in term of some substances consisting mainly of cyclic monomer or hemicellulose from the cotton fabrics. In addition, the mass increment process of CO-PVDF-TiO2-CT obtained between 300 °C and 400 °C, indicates the key role of TiO2 immobilization for thermal stability (Table 2). Afterward, the high weight loss rate of 84.5% at 600 °C, which referred to the hybrids and solid residues of CO-PVDF-TiO2-CT, evidences again the good thermal stability and the uniform distribution of TiO2 within fabric surface. The above results are in good agreement with those obtained by FTIR and SEM analysis. To investigate more details about the char reactivity, the relationship between gasification and surface morphologies are discussed in term of char reaction. As the reaction proceeds, TiO2 particles leave the cotton surface on the outside surface of the char. As a consequence, the TiO2 particles react with CO2 rapidly and then get sintering. The sintering PVDF-TiO2-CT compounds make the cotton fabric interface become less. Therefore, its catalysis is much lower than that in steam gasification. While, the char may become smaller form a PVDF-TiO2-CT layer covering on the surface. The relative PVDF-TiO2-CT interface becomes wide, although some TiO2 nanoparticles get sintering. As result, the PVDF-TiO2-CT layer continues to catalyze the reaction and consequently, a strongest improvement in FR was obtained.
Flame retardancy analysis
The flame retardancy of CO, CO-PVDF-TiO2-CT is evaluated via being exposed directly to a continuing open flame, and the results are reported in Figure 5. A first overview is obtained from that high flammable CO, where the surface was completely burned. This reflects the role of the modification steps, which occurs a visible difference between the prepared composites. Interestingly, the functionalized CO displayed an improvement in the FR properties. As result, the combustion time increased from 41 second to 130.09 second for pristine CO and CO-PVDF-TiO2-CT, respectively. The combustion time of all samples are reported in Table 3. Accordingly, these changes are an interesting signal that evidences the role of our treatment steps. Hence, explanation resides on the strong combination between polymers (i.e. CT and PVDF) and nanocharge (TiO2) that decreases the weight loss and keeps the fabrics structure and the skeleton. Therefore, the PVDF/TiO2/CT combination act as an efficient organic-inorganic composites, which plays the role of barrier to limit by slowing the fire spread, and then the resistances to fire growth improved. Consequently, results on CO-PVDF-TiO2-CT presents good FR properties, as supported by the reduction of the fire spread and the increases of combustion time.
Smoke propagation
After flame retardancy test, the amount of residues obtained on CO-PVDF-TiO2-CT was higher than other counterpart. This result evidences the performance of CO-PVDF-TiO2-CT on char production. The confirmation was obtained from the smoke releases during the combustion time of composites. No visible smoke can be released during the combustion process, suggesting that prepared CO-PVDF-TiO2-CT reduces the propagation of smoke gas. While, the fabric surface was completely coated by char residues. Here, these results may explain the adsorption of smoke in the composite surface without releases. Afterward, the smoke formation was accompanied by an improvement on the FR via the creation of char layer as barrier to limit the fire propagation. As expected, only CO-PVDF-TiO2-CT presents a few smokes as compared to the other materials, indicating the role of chitosan to catalyze FR. This was reflected by the CT/PVDF contained flame retardants, which can interact with free radicals and then limit the flammability of composites [15]. These results were confirmed by the clear decreases of destroyed area, as supported in Table 4. Therefore, the difference of char formation and the obtained residues can visually demonstrate the key role of the post modifications on cotton fabric in their FR properties.
Effect of functionalization steps
According to the results obtained from both characterization and flame retardancy test, the surface modification of cotton fabric was investigated. This was established that CO-PVDF-TiO2-CT exhibits an enhancement of FR properties with a few char residue (Figure 6). Herein, the improvement on fire retardancy must be due to the high capacity of Ti atoms to diffuse or to promote Lewis acid-base interactions with the polymers surface (i.e. CT and PVDF). A possible explanation resides in the formation of carbonates and/or carbonate-like associations with traces of nanocharge and the surrounding lattice oxygen atoms. This must involve multiple interactions, and agrees with the significant enhancement of the hydrophobic character, as confirmed by the contact angle measurements (Scheme 3). Therefore, improvement of fire retardancy is a result of the modification strategies that used during the preparation of composites.
A deep analysis of the FR effect was obtained by measurements of time ignition and the linear propagation velocity (Table 3). Indeed, the required time delay for triggering combustion increases more than three times for CO-PVDF-TiO2-CT, as compared to the untreated cotton fabric. These results evidenced the key role of PET modifications. Accordingly, these data are in agreement with the small-burnt surface upon FR tests (Figure 5).
Thermal degradation mechanism of polymers
According to Gijsman, et al. [30], the main degradation of composites can be obtained by a mechanism of cyclic degradation. In our case, the PVDF and CT polymers chain involved several interaction types with the CO fiber to achieve an equilibrium reaction. From this way, the primary decomposition process will proceed with involving of hydrogen-C-H to nitrogen (N-H) transfer reaction [34-36]. This reaction leading to scission of the chain and the evolution of the fragments terminated by chitosan units. Unlike, the slight tendency to aggregate, titanium dioxide is regularly dispersed by covering the CO. Consequently, adsorption of polymers and creation of a very strong interphase bond between the surface of the metal oxides and the polymer chains (Scheme 2). Therefore, this behavior results in minimal residues production and the thermally and chemically stable CO-PVDF-TiO2-CT, which appeared to be the main factors induce high flame retardancy. This is expected to be even more accentuated due to the simultaneous production of carbon dioxide (CO2), where a possible formation of carbonates and/or carbonate-like interaction with terminal hydroxyls and lattice oxygen atoms belonging to the inorganic moiety. Consequently, the FR ability enhancement may causes by the formation of a protective-char layer hindering oxygen diffusion inside the composite. This barrier should also reduce the flammable mass reservoir and heat transfer, in agreement with previous work [15]. In addition, chitosan may play a certain role in the attenuation of the flammability of the composites [37]. Interestingly, the transfer of degraded products can be limited by these barriers. Then, the carbon oxide or carbon dioxide gases from the prepared composite are released. Here, the reduction in yield of these gases indicates the decrease in fire toxicity and may further lead to the decrease in CO2 amount.
Effect of TiO2 nanocharges
As discussed in the previous sections, nanocharges based on TiO2 are responsible for slowing the combustion of cotton fabrics. The improvement of flame retardancy resulted throughout inhibition of the radical flame propagation reactions in the gas phase. While, the active radicals HO• can be adsorbed at the oxide particles. Therefore, collision energy is transferred to TiO2 and then less reactive HOO• radicals are formed in comparison to those of initial HO•. Afterward, hydrogen-bonding interaction between the carbonyl groups and the hydroxyl groups present on the surface of TiO2 nanocharge are supposed to enhance the thermal stability [34]. Consequently, hydrogen bonds induced a visible slowing on the depolymerization of the polymer chains. In this regard, electrons capture by nanocharges particles act as a gas barrier, preventing the diffusion of degradation products outside the composite (Scheme 4).
Conclusion
Regarding to the high demand for research in the field of composite materials with improved flame retardancy, this work explored an overview of the preparation, functionalization of cotton fabric as efficient candidate with improved thermal stability and FR properties. It was found that flame retardancy was enhanced after the addition of PVDF, TiO2 and CT compounds. The good thermal degradation of the prepared composite evidenced the key role of the functionalization strategies. Results showed that CO-PVDF-TiO2-CT composite decreases the smoke release with low toxicity. Regarding to the above results, our functionalization approach can be used in several applications such textile for environment protection. Thus, new advanced ways for further research for both human and environment safety can be opened.


