
International Journal of Nanoparticles and Nanotechnology
(ISSN: 2631-5084)
Volume 7, Issue 1
Research Article
DOI: 10.35840/2631-5084/5538
Experimental Study on Mineral Phase Structure and Metallurgical Properties of Pellets
Shuangping Yang, Haixing Sun, Tiantian Zhang* and Shouman Liu
Table of Content
Figures
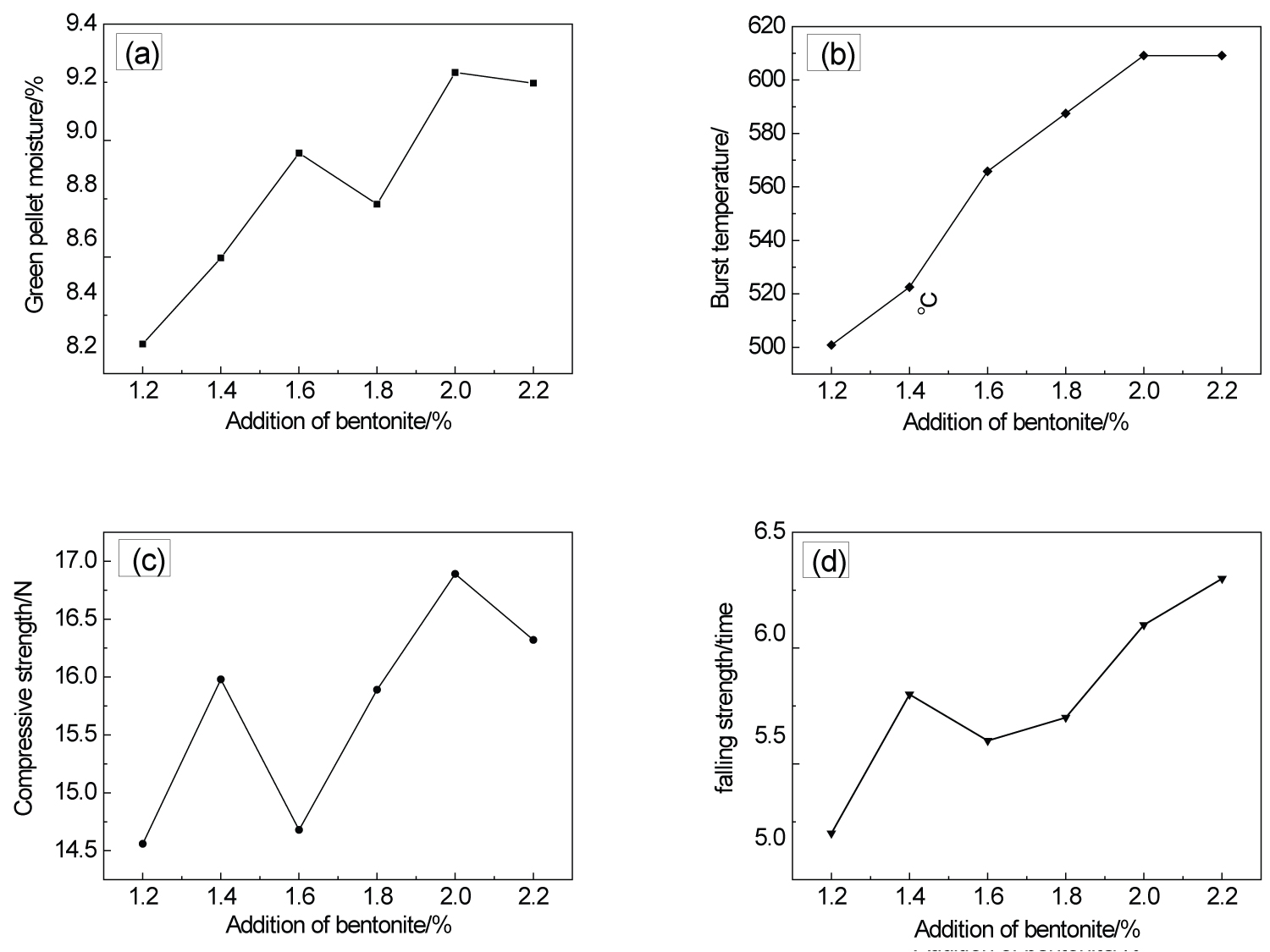
Figure 1: Effect of bentonite on...
Effect of bentonite on quality indices of green pellets. a) Green pellet moisture; b) Burst temperature; c) Compressive strength; d) Falling strength.
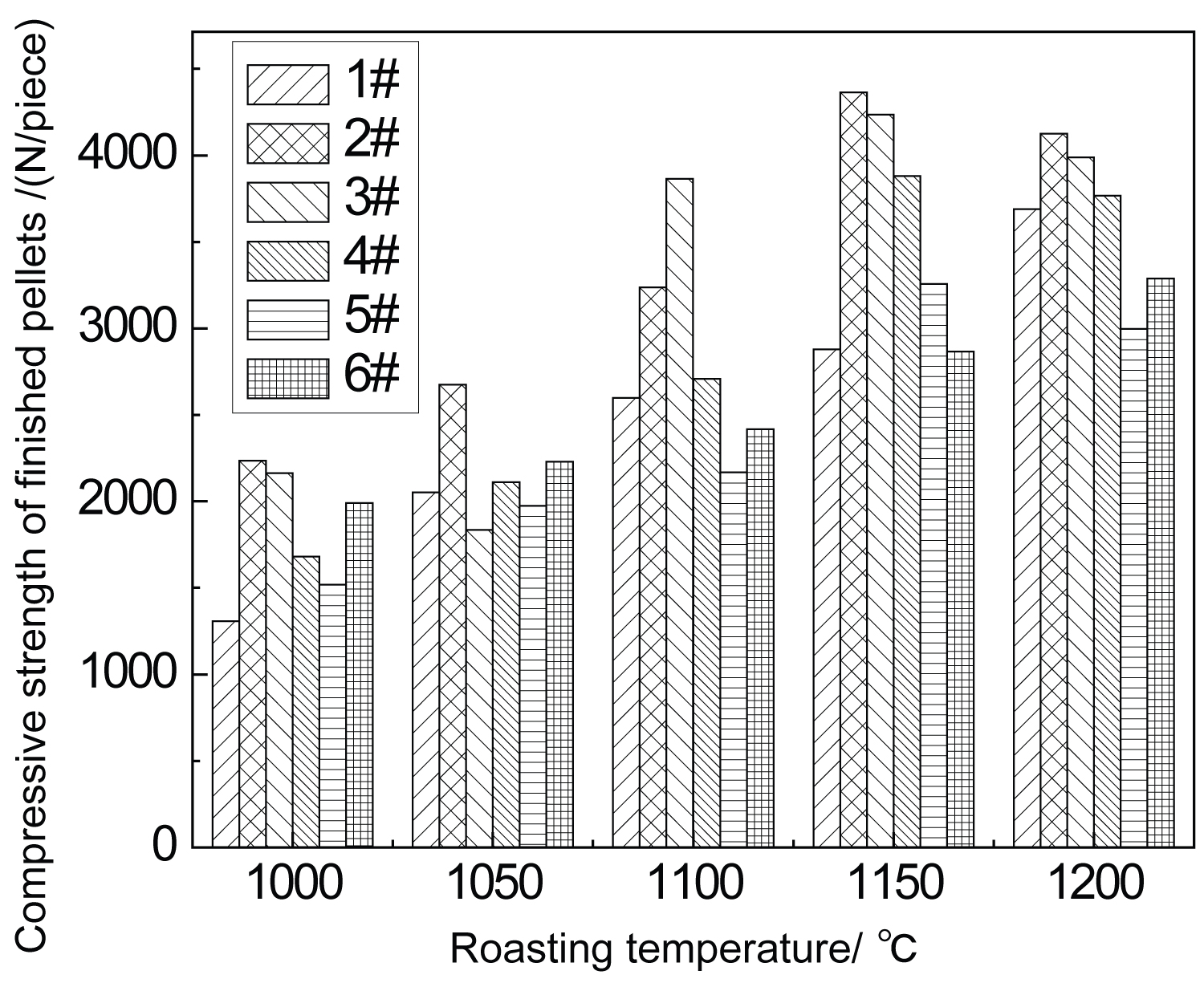
Figure 2: Effect of calcination temperature....
Effect of calcination temperature on compression strength of pellets.
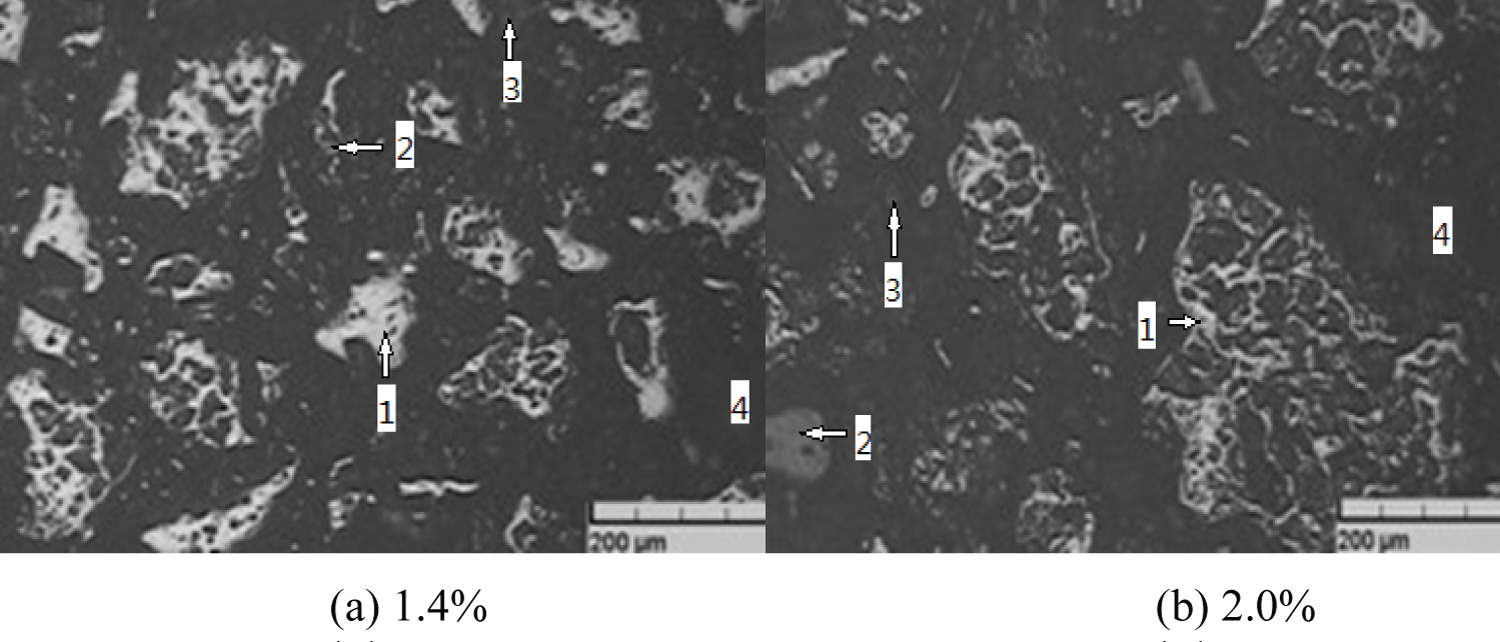
Figure 3: Pellet microstructure....
Pellet microstructure 1) Hematite; 2) Magnetite; 3) Calcium ferrite; 4) Hole.
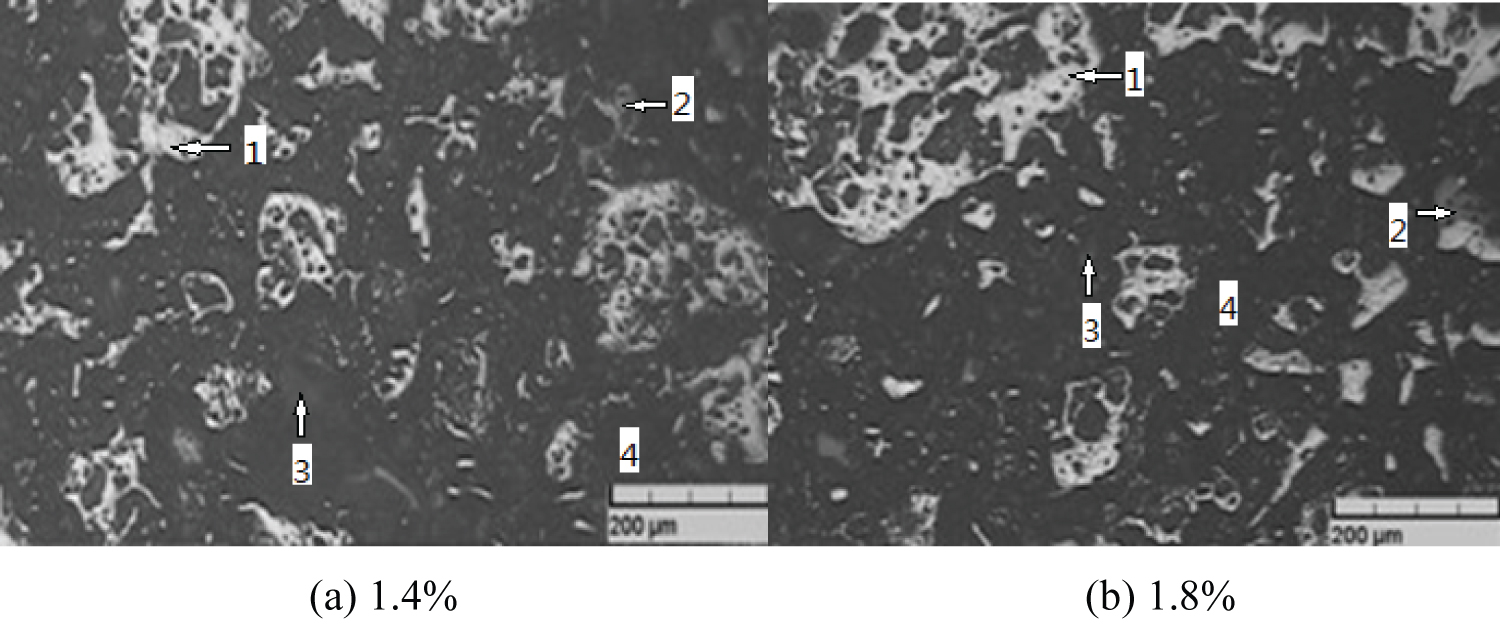
Figure 4: Pellet microstructure....
Pellet microstructure 1) Hematite; 2) Magnetite; 3) Calcium ferrite; 4) Hole.

Figure 5: Pellet microstructure....
Pellet microstructure 1) Hematite; 2) Magnetite; 3) Calcium ferrite; 4) Hole.

Figure 6: Pellet microstructure....
Pellet microstructure 1) Hematite; 2) Magnetite; 3) Calcium ferrite; 4) Hole.

Figure 7: Pellet microstructure...
Pellet microstructure 1) Hematite; 2) Magnetite; 3) Calcium ferrite; 4) Hole.
Tables
Table 1: Chemical composition of iron fines.
Table 2: Particle size composition of iron fines.
Table 3: Chemical composition of bentonite.
Table 4: Bentonite of addition scheme.
References
- Manxing X, Yulan Z (2017) Analysis of pellet technology and production of China in 21st century. Sintering and Pelletizing 42: 25-30.
- Deqing Z, Weiqun H, Congcong Y, Xun H, Jian P (2017) Technology progress of iron ore pellets. Sintered Pellets 42: 42-49.
- Kuangwu Y (2005) About our country's pellet quality. Sintering and Pelletizing 30: 1-4.
- Ma J, Evans DG, Fuller RJ, Stewart DF (2002) Technical efficiency and productivity change of China's iron and steel industry. International Journal of Production Economics 76: 293-312.
- Ming L, Bichun L, Yuanhua Z, et al. (2003) A quadrature experiment study on resulphuring reasons of molten iron inside converter. Research on Iron and Steel 6: 15-18.
- Lingtan K (2003) Technical advances and prospects of raw materials for iron making in China. Iron Making 65: 124.
- Wenquan L (2006) Knowledge and view on development of pellet production in China. Iron Making 3.
- Kuangwu Y (2004) Review of BF burden composition development in EU and the pellet product in our country. Sintering and Pelletizing 29: 42-46.
- Zhaocai W, Shuangyin C, Mansheng C, et al. (2012) Simulation experiment on direct reduction of oxidized pellets of vanadium and titanium-bearing iron concentrates by gas-based shaft furnace. Iron Steel Vanadium Titanium 33: 34-38.
- Jie L, Tielei T, Hongwei X (2014) Study on influence of flux on compressive strength of high magnesium alkaline pellet. Sintering and Pelletizing 39: 36-40.
- Xin J, Gangsheng W, Guo W, et al. (2006) Effect of MgO on Sintering process and metallurgical properties of sinter. Iron & Steel 41: 8-11.
- Wufenglou, Wei W, Heping L, Jianhua P, Zhengliang X, et al. (2017) Micromechanical properties of hematite structure and its influence on pellet strength. Journal of Iron and Steel Research 29: 792-799.
- Tao Y, Qinggele, Li M, Yunqing T, Yan Z, et al. (2020) Analysis of the identification characteristics of the microstructure of pellets. Sintered Pellets 45: 34-38.
- Zhe Z, Xiuli H, Lei L, Xie Z, Dongdong B, et al. (2020) The characteristics of the mineral phase structure of pellets and their influence on metallurgical properties. Journal of North China University of Technology (Natural Science Edition) 42: 7-13.
- Guangquan F (2010) Q&A on pellet production technology. Metallurgical Industry Press, Beijing, China.
Author Details
Shuangping Yang, Haixing Sun, Tiantian Zhang* and Shouman Liu
School of Metallurgical Engineering, Xi'an University of Architecture and Technology, Shaanxi, China
Corresponding author
Tiantian Zhang, School of Metallurgical Engineering, Xi'an University of Architecture and Technology, Xian, 710055, Shaanxi, China, Tel: +86-15129236642.
Accepted: March 29, 2022 | Published Online: March 31, 2022
Citation: Yang S, Sun H, Zhang T, Liu S (2022) Experimental Study on Mineral Phase Structure and Metallurgical Properties of Pellets. Int J Nanoparticles Nanotech 7:038
Copyright: © 2022 Yang S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Pellet is an important part of raw materials for blast furnace. The quality of the pellet directly affects the quality of molten iron. The mineral composition and microstructure of pellets are important factors affecting their metallurgical properties. In order to improve the metallurgical properties of the pellets, this experiment studied the influence of the addition of bentonite and the calcination temperature on the mineral phase structure of the pellets. The optical microscope was used to deeply analyze the root cause of the mineral structure affecting the quality of the pellets, and the best roasting conditions are obtained. The results show that when the addition amount of bentonite is 1.4% and the roasting temperature is 1150 ℃, the Fe2O3 grains are well developed and recrystallized, the grains are closely related, the pores are closed and uniformly distributed, the structure of the pellets is strong and resistant high compressive strength.
Keywords
Pellets, Bentonite, Roasting temperature, Metallurgical properties, Mineral phase structure
Introduction
Pellet is superior to sinter in particle size uniformity, anti-wear and iron content. It is an ideal burden structure for blast furnace [1] to be smelted with high basicity sinter. The low strength and poor reducibility of pellets produced in China affect the effect of blast furnace iron making [2-4]. Most pellets need to be imported [5-8]. Most scholars have studied the influence of roasting process parameters, roasting raw material composition and roasting equipment on the metallurgical properties of pellets [9-11]. However, there are few studies on the influence of mineral composition and microstructure on the quality of pellets.
The mineral composition and microstructure of pellets are closely related to their metallurgical properties [12-14]. In this experiment, the effects of bentonite addition and roasting temperature on the properties and phase structure of pellets were investigated. The phase composition and microstructure of pellets were analyzed by polarizing microscope, and the internal influence rules were revealed, which provided theoretical basis for improving pellet quality and optimizing raw material structure.
Test Materials and Methods
Test materials
The raw materials for this test are iron ore powder and sodium bentonite. The chemical composition and particle size composition of iron ore powder have be shown in Table 1 and Table 2, and the chemical composition of bentonite is shown in Table 3. It can be seen from Table 3 that the main components of bentonite are Montmorillonite Al(SiO4•O10)(OH)2•nH2O, and contain a small amount of quartz, mica, feldspar and kaolinite.
Test method
This experimental ingredient scheme can be found in Table 4.
Firstly, the green pellets were prepared on a disc pelletizer with a diameter of 1200 mm and an inclination of 45° according to the proportioning scheme. During the pelletizing process, the moisture content of the green pellets was controlled at 8~9%. After pelletizing test, the green pellets with diameter of 8~12 mm were selected to test the compressive strength and falling strength, at last, the pellets with different proportions were divided into five groups, which were calcined at 1000 ℃, 1050 ℃, 1100 ℃, 1150 ℃ and 1200 ℃. After the pellets were raised to the preset temperature, they were kept for 1 hour. The representative pellets with good test results were selected and their phase composition and microstructure were analyzed, the optimum amount of bentonite and calcination temperature was found.
Test Results and Analysis
Effect of bentonite on metallurgical quality indices of green pellets
Bentonite is a high-quality additive, which can not only improve the drop strength and compressive strength of the green pellets, but also can regulate the moisture when the moisture of the pellets material is too high, and more importantly, it can increase the burst temperature when the green pellets is dry [15]. In this experiment, the addition of high-quality bentonite can increase the compressive strength by 2.2% under the same drying condition, the relationship between the addition amount and metallurgical properties in Figure 1.
It can be seen from Figure 1a that with the increase of bentonite content, the moisture content of green pellets increases, which is due to the fact that bentonite is easy to disperse in water after encountering water, which enhances the ability of green pellets to keep water. The suitable moisture range of magnetite pellets is 8~9%. So the optimum addition amount of bentonite is about 1.4%.
It can be seen from Figure 1b that the bursting temperature increases with the increase of bentonite content, but the increase gradually slows down, this is because the bentonite acts as a buffer in the drying stage of pellets, which can release water slowly, prevent rapid dehydration, and avoid bursting of pellets due to excessive internal steam pressure. The bursting temperature of green pellets is generally no less than 450 ℃, and all the six groups of tests can meet the production requirements.
It can be seen from Figure 1c and Figure 1d that the compressive strength and falling strength of green pellets are improved to varying degrees with the increase of bentonite addition. This is because montmorillonite in bentonite can promote the formation of liquid phase, and bentonite can change the water content in the pellet, and the water will gather in the pellet, which will reduce the distance between particles and reduce the porosity. On the whole, the six groups of samples can meet the requirements.
Effect of roasting temperature on compressive strength
In the process of pellet roasting, the change of strength reflects the essential change. In order to study the influence of roasting temperature on the compressive strength of pellets after roasting, roasting tests were carried out on six groups of samples, and then the compressive strength of finished pellets was measured. According to the test data, the comparison of compressive strength of pellets at different roasting temperatures was shown in Figure 2.
It can be seen from Figure 2 that the effect of roasting temperature on the compressive strength of pellets is more significant. When the temperature is lower, the compressive strength of each group of samples is generally low, and the effect of roasting consolidation is not achieved. With the increase of roasting temperature, the compressive strength of pellets is improved in different degrees, this is due to the oxidation of magnetite, the growth and recrystallization of Fe2O3 microcrystals. Slag crystallization is formed between pellets through consolidation and diffusion. When the temperature rises to complete oxidation of magnetite, the amount of recrystallization of Fe2O3 will increase, grow and form flakes, when the roasting temperature increases to a certain value, the compressive strength of pellets will not increase anymore.
It can also be found from Figure 2 that the compressive strength of pellets with bentonite addition between 1.4% ~ 1.8% is generally good at each roasting temperature. When the roasting temperature is 1150 ℃, the compressive strength of pellets with bentonite ratio of 1.4%, 1.6% and 1.8% reaches the maximum value. Considering the economic cost, it is better to choose 1.4% bentonite.
Mineral Phase Analysis of Pellet
The metallurgical performance of pellet is the external characterization of the difference of ore phase structure. In order to study the relationship between the ore phase structure and the metallurgical properties of pellets, the ore phase of representative pellets was detected. The samples selected in this experiment are: 1000 ℃, 1050 ℃, 1100 ℃, 1150 ℃, 1200 ℃ in 2 ore blending scheme; 1100 ℃, 1200 ℃ in 3 ore blending scheme; 1050 ℃, 1150 ℃ in 4 ore blending scheme; 1000 ℃, 1100 ℃, 1200 ℃ in 5 ore blending scheme. Each group of samples was magnified 200 times under XJP-6A type polarizing microscope and then photographed for mineral composition and microstructure analysis. The analysis is as follows:
(1) At 1000 ℃, the microstructure of two kinds of pellets with bentonite ratio of 1.4% and 2.0% is shown in Figure 3.
It can be observed from Figure 3 that due to the low roasting temperature, magnetite is not completely oxidized, and the crystallization of Fe2O3 is not perfect, resulting in more individual particles of Fe2O3 recrystallization. The grain development of Fe2O3 in Figure 3a is more perfect and evenly distributed than that in Figure 3b, and the structure of Figure 3a is relatively dense, the pellet strength is also higher, and the structure of Figure 3b is loose. The results show that Fe2O3 grain is still in the growth stage, there is no crystal connection and the pore size is different, so the pellet strength is also low.
(2) At 1050 ℃, the microstructure of two kinds of pellets with bentonite ratio of 1.4% and 1.8% is shown in Figure 4.
It is observed from Figure 4 that the grain growth angle of Fe2O3 in Figure 4b is better than that in Figure 4a, but the grains are not related to each other, and there are many single grains, and the structure is loose and the porosity is high. In Figure 4a, some Fe2O3 grains begin to connect with each other and the shape is regular, so the strength is higher, but die grains are still in the growth stage.
(3) The microstructure of three kinds of pellets with bentonite ratio of 1.4%, 1.6% and 2.0% at 1100 ℃ is shown in Figure 5.
It is observed from Figure 5 that the Fe2O3 grains in Figure 5a are uniformly distributed and closely connected, and the grains are well developed and the crystal form is coarse. The grains in Figure 5b and Figure 5c are weakened. Only a few grains in Figure 5c develop and grow, and no large areal intergranular grains are formed. The recrystallization of most Fe2O3 grains is still in the development stage, and the porosity is different. In some areas, Fe3O4 is not oxidized completely, and the grain angle is obvious.
(4) At 1150 ℃, the microstructure of two kinds of pellets with bentonite ratio of 1.4% and 1.8% is shown in Figure 6.
It is observed from Figure 6 that hematite grains in the two pellets have been closely related to each other and become interconnected crystals. The grains are coarse and closely connected, which shows that the structural force of pellets is strong and the compressive strength is high. Compared with Figure 6b, the size of holes in Figure 6a is not uniform, and some grains still need to be interconnected. Figure 6b is a whole interconnected crystal with closed pores. Therefore, the pellet has high degree of consolidation and high strength.
(5) The microstructure of two kinds of pellets with bentonite ratio of 1.4%, 1.6% and 2.0% at 1200 ℃ is shown in Figure 7.
It can be observed from Figure 7 that the Fe2O3 grains in Figure 7a have completely become intergranular crystals, and the pellet strength has been greatly improved; while the morphology of Figure 7b and Figure 7c are similar. Although the grains have been completely connected, a small part of Fe2O3 grains need to be fully oxidized and recrystallized to form interconnected crystals. However, the pellets in Figure 7b and Figure 7c still have high strength.
To sum up, the law of pellet consolidation change can be obtained. That is, the effect of pellet roasting and consolidation is gradually improved with the increase of temperature. The higher the roasting temperature is, the more complete the green pellet oxidation is, and the higher the pellet strength is. And at the same roasting temperature, the consolidation effect of the sample with bentonite ratio of 1.4% is better.
Conclusion
1) The addition of bentonite can increase the moisture content and bursting temperature of green pellets, and improve the compressive strength and falling strength of green pellets in varying degrees. Increasing the roasting temperature can obviously improve the compressive strength of pellets, but when the temperature is higher than 1150 ℃, the compressive strength will decrease.
2) Through roasting test and phase analysis of pellet, the optimum bentonite ratio and roasting temperature for pellet production arc determined: Bentonite ratio is 1.4%, roasting temperature is 1150 ℃.
3) The oxidation of Fe3O4 to Fe2O3 grains is a process of growth and development. Fe2O3 grains recrystallize and interconnect to form a whole for consolidation. The degree of consolidation gradually improves with the increase of temperature, and the strength of corresponding pellets also increases.

