International Journal of Optics and Photonic Engineering
(ISSN: 2631-5092)
Volume 6, Issue 2
Research Article
DOI: 10.35840/2631-5092/4540
Dye-Sensitized Solar Cell (DSSC) Applications based on Cyano Functional Small Molecules Dyes
Mustafa Can1, Ali Kemal Havare2* and Ender Akan3
Table of Content
Figures
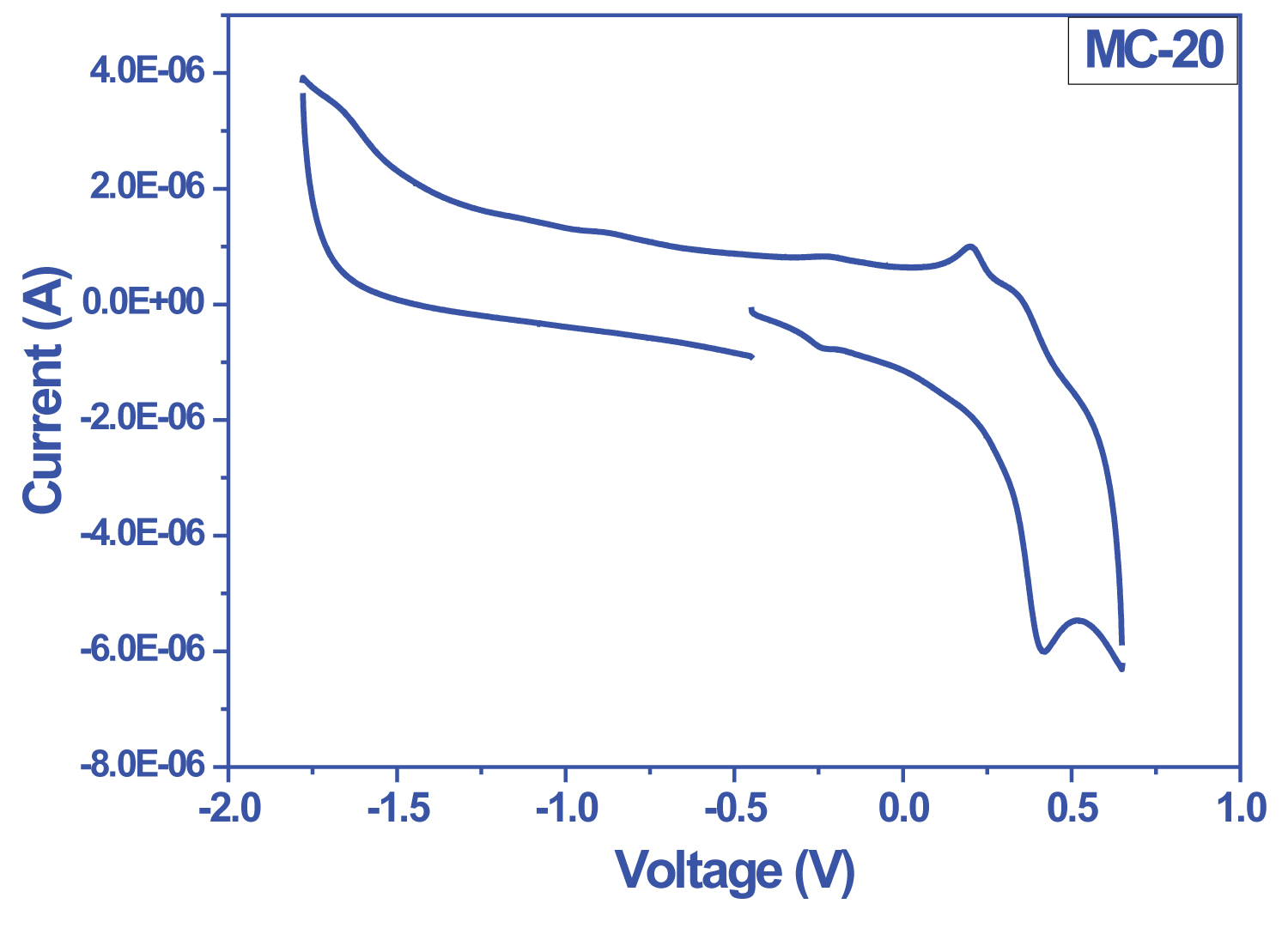
Figure 1: Cyclic voltomogram of MC-20....
Cyclic voltomogram of MC-20 taken in 0.1 M TBAHFF-DMF at 100 mVs-1 scanning speed.
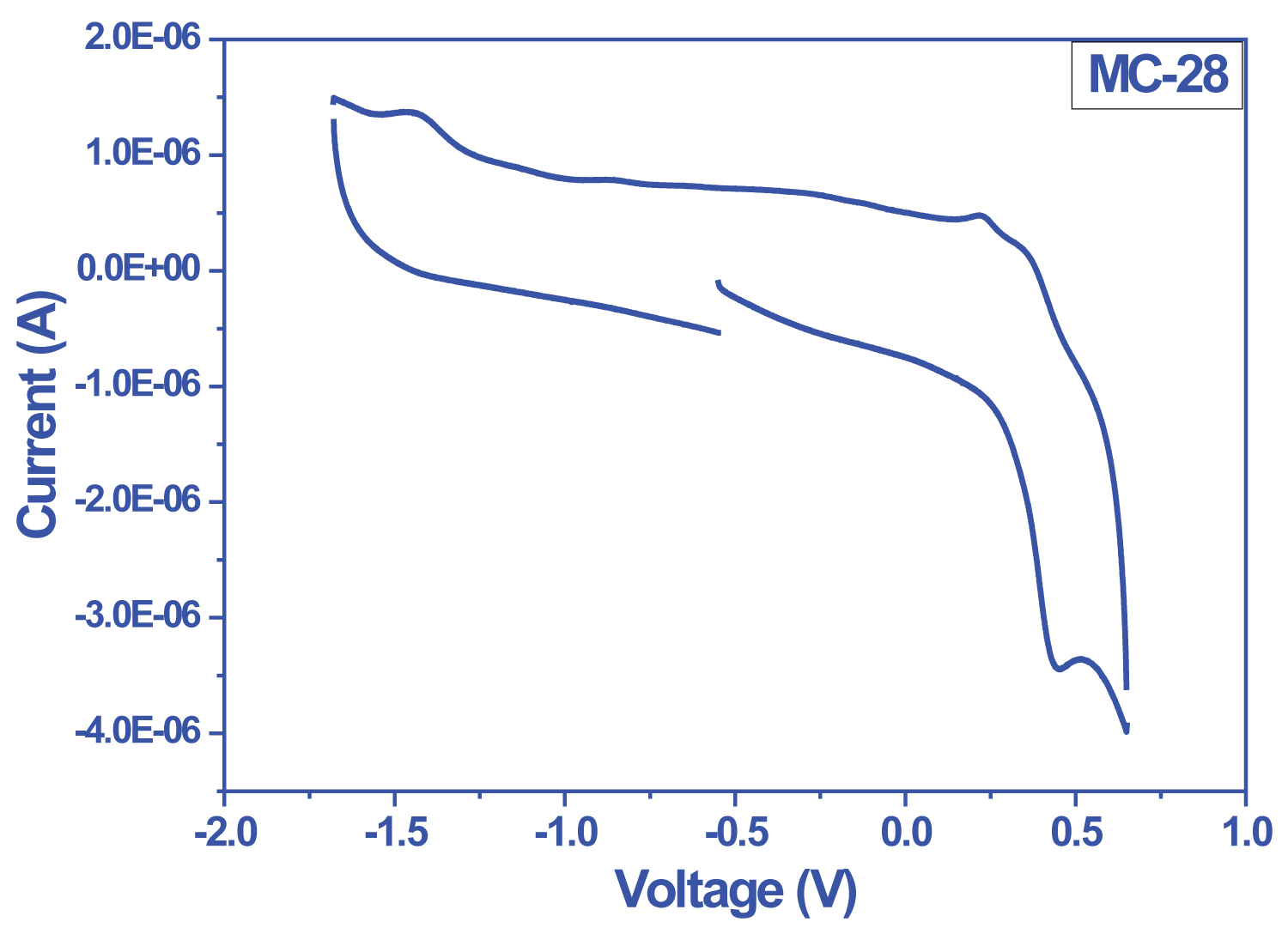
Figure 2: Cyclic voltomogram of MC-28.....
Cyclic voltomogram of MC-28 taken in 0.1 M TBAHFF-DMF at 100 mVs-1 scanning speed.
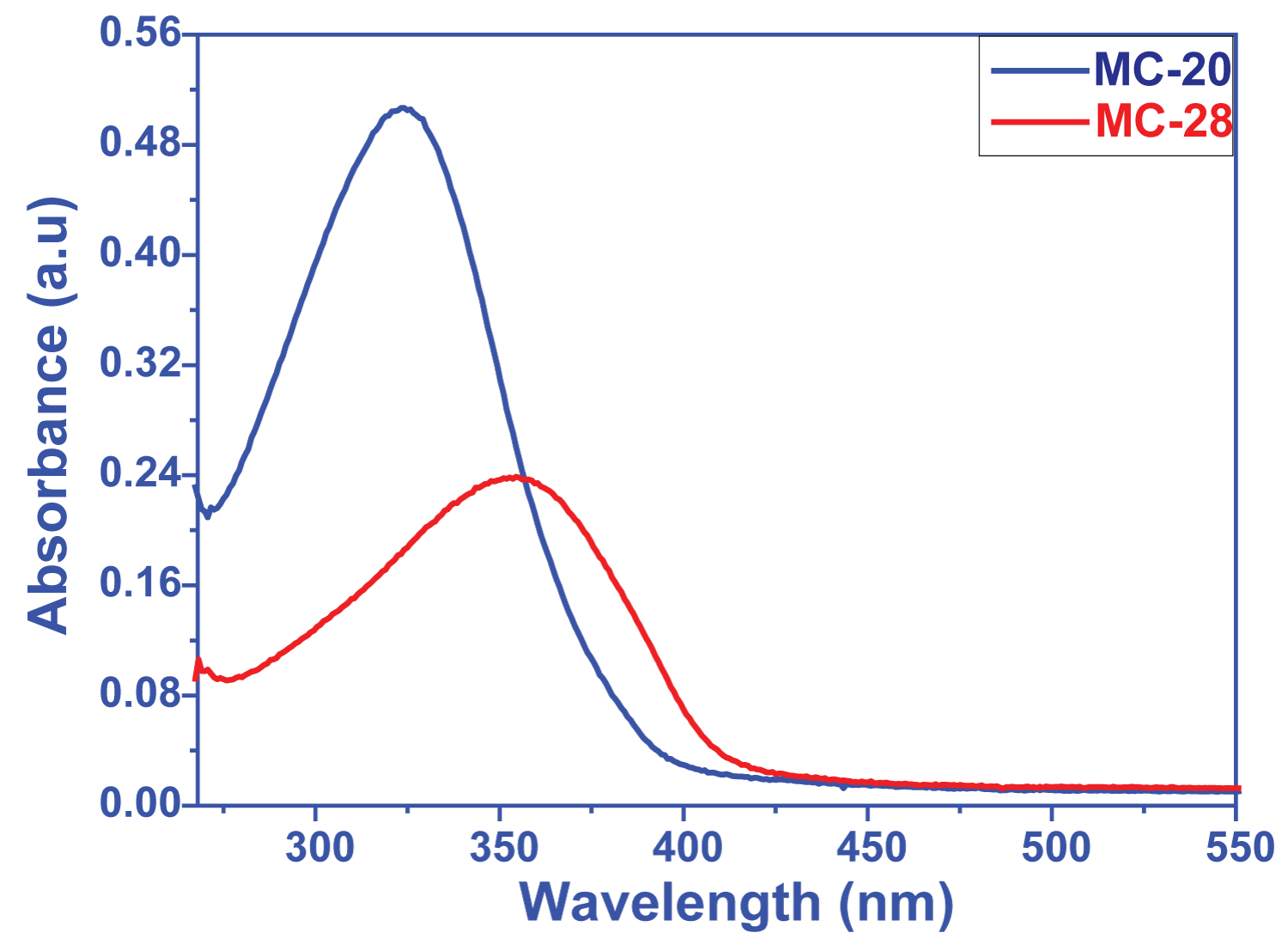
Figure 3: UV-vis absorption spectra of....
UV-vis absorption spectra of the dyes MC20 and MC28 in DMSO solution.
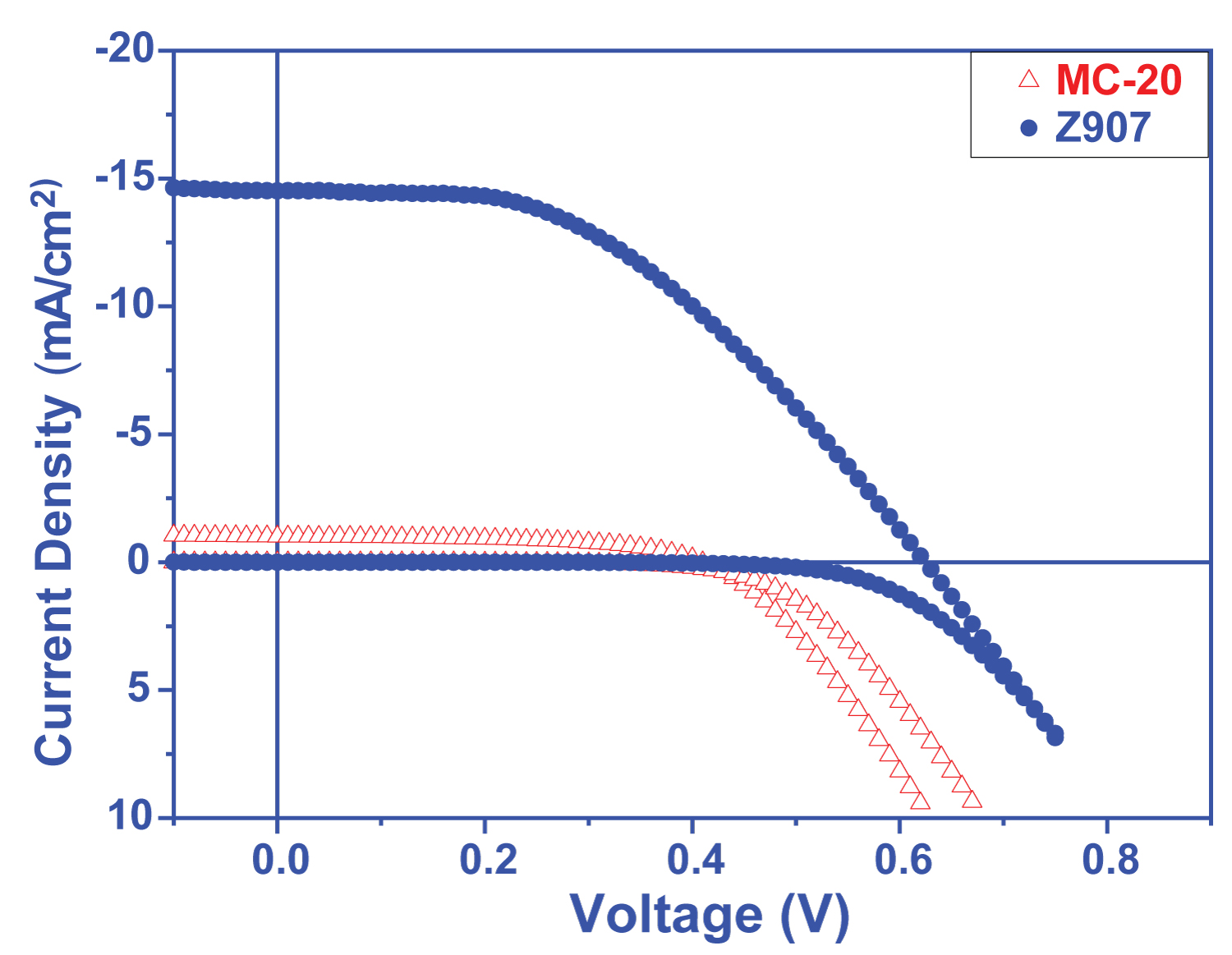
Figure 4: Current density - voltage....
Current density - voltage graphs of solar cells prepared from MC20 dye.
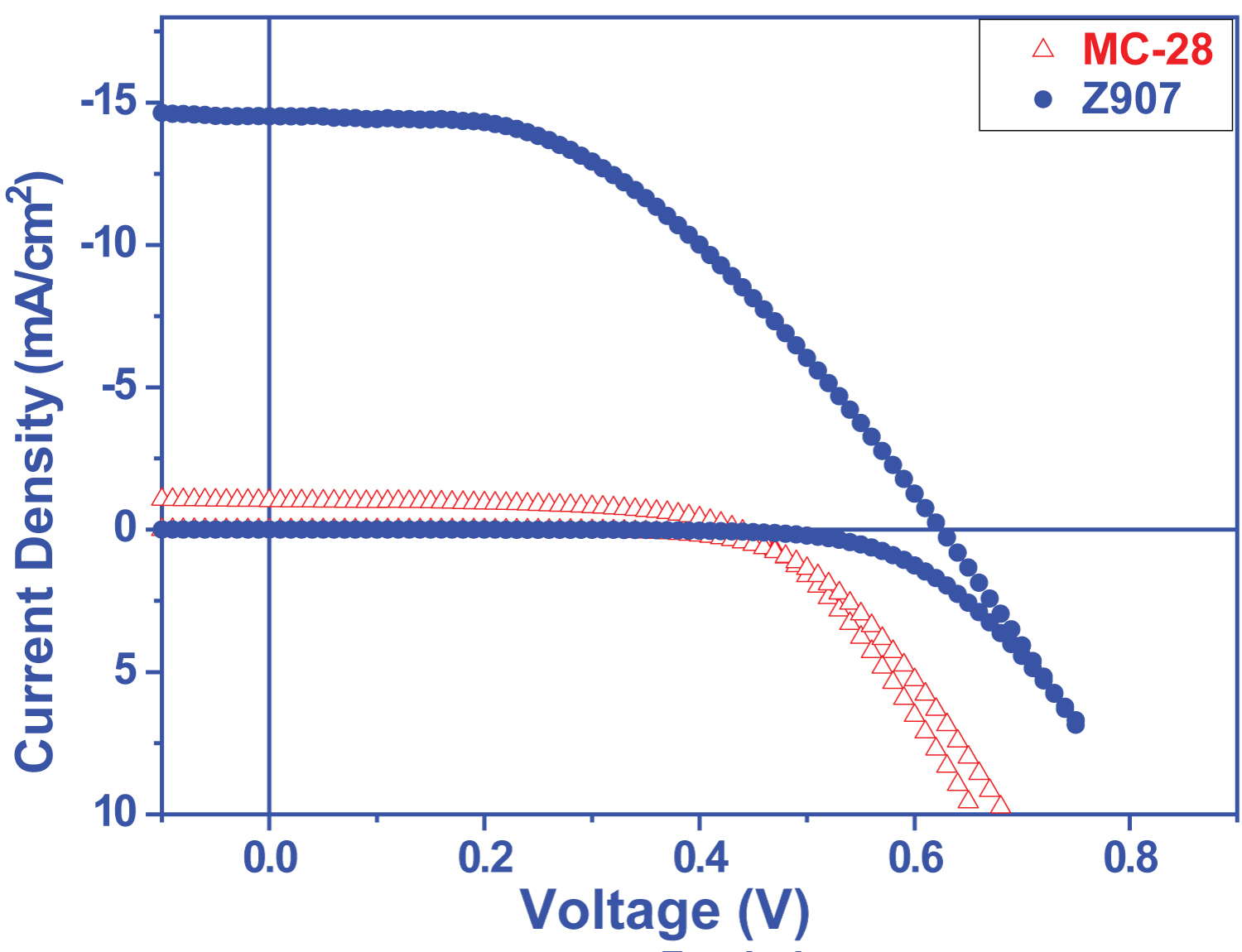
Figure 5: Current density - voltage....
Current density - voltage graphs of solar cells prepared from MC28 dye.

Scheme 1: General representation of....
General representation of the dye whose molecular structure is designed as D-π-A.
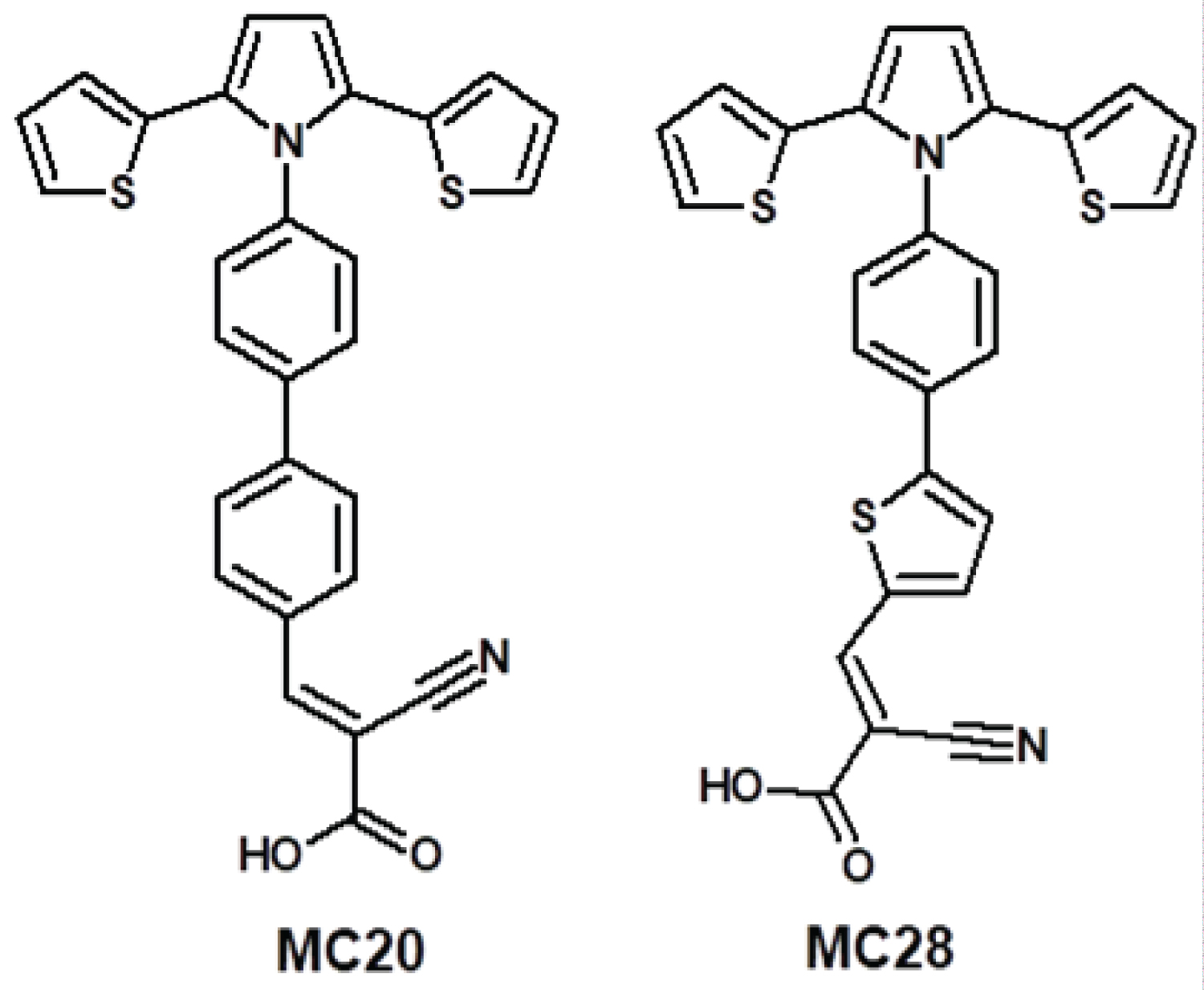
Scheme 2: The synthesized small molecules.....
The synthesized small molecules (2E) -2-Cyano-3- [4 '- (2,5-di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-yl] acrylic acid (MC-20). (2E) -2-Cyano-3- 5- [4- (2,5-di-2-thienyl-1H-pyrrol-1-yl) phenyl] -2-thienyl acrylic acid (MC-28).
Tables
Table 1: The Highest Occupied Molecular Orbital (HOMO) - The Lowest Unoccupied Molecular Orbital (LUMO) of the dyes and their wavelength absorption.
Table 2: Photovoltaic efficiency obtained from DSSCs prepared using MC20 and MC28 dyes.
References
- Servaites JD, Ratner MA, Marks TJ (2011) Organic solar cells: A new look at traditional models. Energy & Environmental Science 4: 4410-4422.
- Swami R (2012) Solar cell. International Journal of Scientific and Research Publications 2: 1-5.
- Kaltenbrunner M, White MS, Glowacki ED, Sekitani T, Someya T, et al. (2012) Ultrathin and lightweight organic solar cells with high flexibility. Nat Commun 3.
- Mazhari B (2006) An improved solar cell circuit model for organic solar cells. Solar Energy Materials and Solar Cells. 90: 1021-1033.
- Sinke WC, Wienk MM (1998) Solid-state organic solar cells. Nature 395: 544-545.
- Kalowekamo J, Baker E (2009) Estimating the manufacturing cost of purely organic solar cells. Solar Energy 83: 1224-1231.
- Gong J, Liang J, Sumathy K (2012) Review on dye-sensitized solar cells (DSSCs): Fundamental concepts and novel materials. Renewable and Sustainable Energy Reviews 16: 5848-5860.
- Gong J, Sumathy K, Qiao Q, Zhou Z (2017) Review on dye-sensitized solar cells (DSSCs): Advanced techniques and research trends. Renewable and Sustainable Energy Reviews 68: 234-246.
- Semalti P, Sharma SN (2020) Dye sensitized solar cells (DSSCs) electrolytes and natural photo-sensitizers: A review. J Nanosci Nanotechnol 20: 3647-3658.
- Karim NA, Mehmood U, Zahid HF, Asif T (2019) Nanostructured photoanode and counter electrode materials for efficient Dye-Sensitized Solar Cells (DSSCs). Solar Energy 185: 165-188.
- Kumar R, Sahajwalla V, Bhargava P (2019) Fabrication of a counter electrode for dye-sensitized solar cells (DSSCs) using a carbon material produced with the organic ligand 2-methyl-8-hydroxyquinolinol (Mq). Nanoscale Adv 1: 3192-3199.
- Haque SA, Park T, Xu C, Koops S, Schulte N, et al. (2004) Interface engineering for solid-state dye-sensitized nanocrystalline solar cells: The use of ion-solvating hole-transporting polymers. Advanced Functional Materials 14: 435-440.
- Cavallo C, Di Pascasio F, Latini A, Bonomo M, Dini D (2017) Nanostructured semiconductor materials for dye-sensitized solar cells. Journal of Nanomaterials 2017.
- Agarwala P, Kabra D (2017) A review on triphenylamine (TPA) based organic hole transport materials (HTMs) for dye sensitized solar cells (DSSCs) and perovskite solar cells (PSCs): Evolution and molecular engineering. J Mater Chem A 5: 1348-1373.
- Ammar AM, Mohamed HS, Yousef MM, Abdel-Hafez GM, Hassanien AS, et al. (2019) Dye-sensitized solar cells (DSSCs) based on extracted natural dyes. Journal of Nanomaterials 2019.
- Heredia D, Natera J, Gervaldo M, Otero L, Fungo F, et al. (2010) Spirobifluorene-bridged donor/acceptor dye for organic dye-sensitized solar cells. Org Lett 12: 12-15.
- Click KA, Beauchamp DR, Garrett BR, Huang Z, Hadad CM, et al. (2014) A double-acceptor as a superior organic dye design for p-type DSSCs: high photocurrents and the observed light soaking effect. Physical Chemistry Chemical Physics 16: 26103-26111.
- El-Shishtawy RM, Asiri AM, Aziz SG, Elroby SAK (2014) Molecular design of donor-acceptor dyes for efficient dye-sensitized solar cells I: a DFT study. J Mol Model 20: 2241.
- Zhang J, Kan Y-H, Li H-B, Geng Y, Wu Y, et al. (2012) How to design proper p-spacer order of the D-p-A dyes for DSSCs? A density functional response. Dyes and Pigments 95: 313-321.
- Kim BG, Chung K, Kim J (2013) Molecular design principle of all-organic dyes for dye-sensitized solar cells. Chemistry 22: 5220-5230.
- Michinobu T, Satoh N, Cai J, Li Y, Han L (2014) Novel design of organic donor-acceptor dyes without carboxylic acid anchoring groups for dye-sensitized solar cells. J Mater Chem C 2: 3367-3372.
- Slimi A, Hachi M, Fitri A, Benjelloun AT, Elkhattabi S, et al. (2020) Effects of electron acceptor groups on triphenylamine-based dyes for dye-sensitized solar cells: Theoretical investigation. Journal of Photochemistry and Photobiology A: Chemistry 398: 112572.
- Hachi M, Slimi A, Fitri A, Benjelloun AT, Khattabi SEI, et al. (2021) Theoretical design and characterization of D-A1-A based organic dyes for efficient DSSC by altering promising acceptor (A1) moiety. Journal of Photochemistry and Photobiology A: Chemistry 407: 113048.
- Cho MJ, Park SS, Yang YS, Yang YS, Kim JH, et al. (2010) Molecular design of donor-acceptor-type cruciform dyes for efficient dyes-sensitized solar cells. Synthetic Metals 160: 1754-1760.
- Wu H, Xie X, Zhang J, Li S, Shen Z, et al. (2020) Low-energy-gap organic photosensitizers with phenalenothiophene and benzoindenothiophene as primary electron-donors for durable dye-sensitized solar cells. Journal of Power Sources 451: 227748.
- Ji J-M, Zhou H, Kim HK (2018) Rational design criteria for D-p-A structured organic and porphyrin sensitizers for highly efficient dye-sensitized solar cells. J Mater Chem A 6: 14518-14545.
- Rashid MAM, Hayati D, Kwak K, Hong J (2019) Computational investigation of tuning the electron-donating ability in metal-free organic dyes featuring an azobenzene spacer for dye-sensitized solar cells. Nanomaterials (Basel) 9: 119.
- Shimogawa H, Endo M, Taniguchi T, Nakaike Y, Kawaraya M, et al. (2017) D-p-A dyes with an intramolecular B-N coordination bond as a key scaffold for electronic structural tuning and their application in dye-sensitized solar cells. Bull Chem Soc Jpn 90: 441-450.
- Cui B, Yan L, Gu H, Yang Y, Liu X, et al. (2018) Fluorescent carbon quantum dots synthesized by chemical vapor deposition: An alternative candidate for electron acceptor in polymer solar cells. Optical Materials 75: 166-173.
- Chen X, Liao Y, Liu Y, Zhu C, Chen TQ, et al. (2017) Dye sensitizers of polymer using the complex of Cd (II) or Cu (II) with imidazole as auxiliary electron acceptor for dye-sensitized solar cells. Dyes and Pigments 139: 420-430.
- Barea EM, Zafer C, Gultekin B, Aydin B, Koyuncu S, et al. (2010) Quantification of the effects of recombination and injection in the performance of dye-sensitized solar cells based on N-substituted carbazole dyes. The Journal of Physical Chemistry C 114: 19840-19848.
- Zafer C (2006) Organik boya esasli nanokristal yapili ince film günes pili üretimi. Ege Üniversitesi, Turkey.
Author Details
Mustafa Can1, Ali Kemal Havare2* and Ender Akan3
1Department of Engineering Sciences, Izmir Katip Celebi University, Izmir, Turkey
2Electric Electronics Engineering, Photoelectronics Lab (PEL), Toros University, Mersin, Turkey
3Materials Science and Engineering, Faculty of Dentistry, Izmir Katip Celebi University, Izmir, Turkey
Corresponding author
Ali Kemal Havare, Electric Electronics Engineering, Photoelectronics Lab. (PEL), Toros University, Mersin, Turkey.
Accepted: September 01, 2021 | Published Online: September 03, 2021
Citation: Can M, Havare AK, Akan E (2021) Dye-Sensitized Solar Cell (DSSC) Applications based on Cyano Functional Small Molecules Dyes. Int J Opt Photonic Eng 6:040
Copyright: © 2021 Can M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
This work focused on synthesizing of the electro active two small molecules ((2E)-2-Cyano-3-[4'-(2,5-di-2 thienyl-1H-pyrrol-1-yl) biphenyl-4-yl] acrylic acid (MC-20) (2E) -2-Cyano-3-5-[4-(2,5-di-2-thienyl-1H pyrrol-1 yl) phenyl] -2-thienyl acrylic acid (MC-28)) consisting of planar molecule part with different absorptions-emissions energy levels and applications of these small molecules in DSSC device to understand their performance. The optical properties of MC-20 and MC-28 were determined by UV-Vis absorption and fluorescence measurements, and electrochemical properties were illuminated by cyclic voltammetry (CV). The 1H-NMR spectroscopy is used to characterize the molecular structure of the synthesized materials. The obtained electrochemical and optical data were used to calculate Highest Occupied Molecular Orbital-Lowest Unoccupied Molecular Orbital (HOMO-LUMO) band gaps. As a result, these synthesized molecules have been used in Dye sensitized solar cells (DSSCs). The performance of these materials was analyzed by calculating the efficiency of the DSSC device. Obviously, it can be said that linking different donor atom S to the molecule has a little positive effect on photovoltaic efficiency.
Keywords
Donor-acceptor dye materials, DSSCs, Cell efficiency
Introduction
The use of existing fossil fuels for energy needs is accepted as the main cause of global climate change. Silicon solar cells or organic solar cell which are an alternative solution to this traditional fossil energy and can directly converts solar energy to electrical energy, have become the focus of attention [1-5]. The limited performance and high production costs of inorganic silicon-based solar cells used today have increased the search for alternative materials to these cells [6]. DSSC technology is an alternative to silicon-based solar cells technology [7-9]. The DSSC is constituted of three main parts. These layers can be listed as the two conductive contacts, the redox layer and the dye material that consisting of organic semiconductor material located between these contacts [8-11]. In this point organic semiconductor materials play an important role in layer of the DSSC device [12-15]. One of the layers that the molecular structure of the organic semiconductor dye material can be designed to consist of donor and acceptor parts [16-18]. Thus, this design let to be transferring the electrons easily in the device. In order for all molecular electronic technology devices to work efficiently, the electron transfer in organic semiconductor dye between the electron donor (donor) and electron acceptor (acceptor) parts must take place effectively [19-22]. Donor-Acceptor type organic semiconductor materials (small molecules) are of great interest, especially since they are systems in which energy and electron transfer can be controlled [16,23-25]. One of the advantages of these materials is that the p-doping and n-doping properties of the material are determined by controlling the energy of the conjugated π system with the regular repetition of donor and acceptor monomers in the chain, and the HOMO-LUMO band gap can be easily tuned [19,26-28]. Because of these properties, it is common to use the donor-acceptor type materials in molecular electronics, nonlinear optical materials, and optoelectronic technology devices. Donor-Acceptor type small molecules are widely used in organic electronics, especially since they are systems in which energy and electron transfer can be controlled. One of the advantages of these materials is that the p-doping and n-doping properties of the material are determined by controlling the energy of the conjugated pi system with the regular repetition of donor and acceptor monomers in the chain, and the HOMO-LUMO band gap can be easily tuned. Because of these properties, the use of donor-acceptor type materials in molecular electronics, nonlinear optical materials, and optoelectronic technology devices is common [20,29,30]. Organic semiconductor materials-based solar cells continue to be studied intensively due to their low production cost, their ability to cover large surfaces, their mechanical flexibility, and their constantly increasing efficiency. In addition, since organic solar cells can be produced in transparent form, it offers the opportunity to use this technology in all areas of daily life such as automotive and construction glass.
In this study, different types of donor-acceptor molecule-based multi-functional materials that will add innovation to optoelectronic technology and have stable and high efficiency in device applications were synthesized and DSSC was applied. Molecule centered donor-acceptor type organic semiconductor material with planar geometry was synthesized and the molecular structure of this material was analyzed by 1H-NMR techniques. The absorbance spectrum of these molecules was measured with the UV-Vis technique, and their electrochemical properties were determined by cyclic voltammetry. Finally, the band gap values of these materials were obtained by calculating HOMO-LUMO values and they were used as dye in DSSC to calculate the efficiency of the solar cell.
Material and Methods
Synthesis of the dyes and solar cell application
The properties of organic dyes can be changed by attaching different groups to the molecule. With a correct design, efficiency improvement studies can be carried out in DSSC cells by changing the wavelength, damping coefficient and energy levels of the dye. Organic dyes have many advantages compared to transition-metal based dyes, such as high damping, easier modification and less environmental damage. However, organic dyes have problems such as narrow absorption band gap, tendency to aggregate and stability.
The common system in the design of an organic dye used in DSSC cells is molecules in the D-π- A order. As shown in Scheme 1, molecules generally need to be synthesized in such a way that electron donor (donor), conjugate system (-π) and electron acceptor are formed. For DSSC cells of this structure, the material can give the best result for charge separation after stimulation. Usually, the common strategy is to determine which group contributes more to the efficiency of the solar cell by replacing one of the above-mentioned D-π-A structures.
The synthetic path followed for MC20 and MC28 dyes (Scheme 2) synthesized for DSSC applications:
(2E) -2-Cyano-3-[4'-(2,5-di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-yl] acrylic acid synthesis synthesis (MC-20)
First Stage: Synthesis of 1,4-Di-2-thienylbutane-1,4-dione
AlCl3 (16 g; 0.12 mol) was added in 50 ml of CH2Cl2 under argon atmosphere and the ambient temperature was cooled to 0 ℃. Then the solution of thiophene (9.61 ml; 0.12 mol) and succinyl dichloride compound (5.51 ml; 0.05 mol) in 15 ml CH2Cl2 was added drop wise to the medium. The medium was observed to turn red in color and this mixture was stirred for 18 hours at room temperature. At the end of the period, the reaction mixture was poured into 100 ml of ice water containing 5 ml of HCl and stirred for a further 2 hours (the solution was observed to turn green in color). The reaction mixture was washed with 2M HCl, water and NaHCO3, respectively, and dried with MgSO4. The crude product, obtained in green color by removal of CH2Cl2, was purified by crystallization with EtOH (white product).
1,4-Di-2-thienylbutane-1,4-dione; [MZ-14], 1H NMR (CDCl3): 7.80 (dd, 2H), 7.63 (dd, 2H), 7.13 (dd, 2H), 3.38 (s, 4H)
Second Stage: Synthesis of 1- (4-Bromophenyl) -2,5-di-2-thienyl-1H-pyrrole
In a single neck flask, 1,4-di-2-thienylbutane-1,4-dione (0.5 g; 2 mmol), p-bromoaniline (0.82 g; 2.2 mmol) and p-toluenesulfonic acid (6.8 mg; 0.4 mmol) 60 ml of dry toluene were refluxed using the "Dean Stark" trap. The reaction followed by TLC method was continued until the starting compounds were consumed in the medium. At the end of the reaction, toluene was removed under vacuum. The product purified by column chromatography (silica gel, CH2Cl2) was dried in a vacuum oven (50 ℃) (light yellow solid).
1- (4-Bromophenyl) -2,5-di-2-thienyl-1H-pyrrole, 1H NMR (CDCl3): 7.57 (d, 2H), 7.20 (d, 2H), 7.12 (d, 2H), 6.88 (t, 2H), 6.58 (d, 2H), 6.55 (s, 2H).
Third Stage: Synthesis of 4 '- (2,5-Di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-carbaldehyde
1-(4-bromophenyl)-2,5-di-2-thienyl-1H-pyrrole (78 mg; 0.2 mmol), (4-formylphenyl) boronic acid (30) in anhydrous DME (12 ml) in a two-necked flask. mg; 0.2 mmol), Pd (PPH3)4 (16 mg; 0.013 mmol) were added. The reaction temperature was gradually increased and when it reached 50 ℃, 2 mL of K2CO3 (1 M) was added to the reaction medium and the mixture was stirred at 90 ℃ for 30 hours. At the end of the process, water (25 ml) and CH2Cl2 (25 ml) were added to the reaction mixture cooled to room temperature and extracted twice. The organic phases were combined and the organic solvent was evaporated. The product purified by column chromatography (silica gel; CH2Cl2:hexane 1:1) was dried (yellow solid) in a vacuum oven (50 ℃).
4'-(2,5-Di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-carbaldehyde, 1H NMR (CDCl3): 10.10 (s, 1H), 8.01 (d, 2H), 7.96 (d, 2H), 7.73 (d, 2H), 7.44 (d, 2H), 7.10 (d, 2H), 6.86 (t, 2H) 6.61 (d, 2H), 6.58 (s, 2H).
Fourth Stage: Synthesis of (2E) -2-Cyano-3- [4 '- (2,5-di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-yl] acrylic acid.
4 '- (2,5-di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-carbaldehyde (83 mg; 0.2 mmol) and cyanoacetic acid (17 mg; 0.2 mmol) in acetonitrile a catalytic amount of piperidine was added to the solution. The reaction mixture was boiled at 90 ℃ for 28 hours. Purified water (40 mL) was added to the mixture cooled to room temperature and extracted with CHCl3 (40 mL). The combined organic phases were washed again with pure and brine solution. The resulting organic phase was dried over dry MgSO4. After evaporation of the organic solvent, the crude product obtained was purified (reddish solid) by column chromatography (silica gel, CH2Cl2:MeOH, 3:1) method.
(2E) -2-Cyano-3- [4 '- (2,5-di-2-thienyl-1H-pyrrol-1-yl) biphenyl-4-yl] acrylic acid, 1H NMR (DMSO): 8.11 (s, 1H), 8.04 (d, 2H), 7.95 (d, 2H), 7.93 (d, 2H), 7.45 (d, 2H), 7.30 (d, 2H), 6.89 (t, 2H) 6.72 (d, 2H), 6.59 (s, 2H); FT-IR (KBr, cm-1): -OH, 3442; (C≡N, nitrile), 2337; C = 0.1622; C = C-H, 3040; -C=C-, 1600; (C=C, Ph) 1506 and 1496.
The synthesis detail and the characterization (1H NMR) is given in the data sheet at the end of the paper.
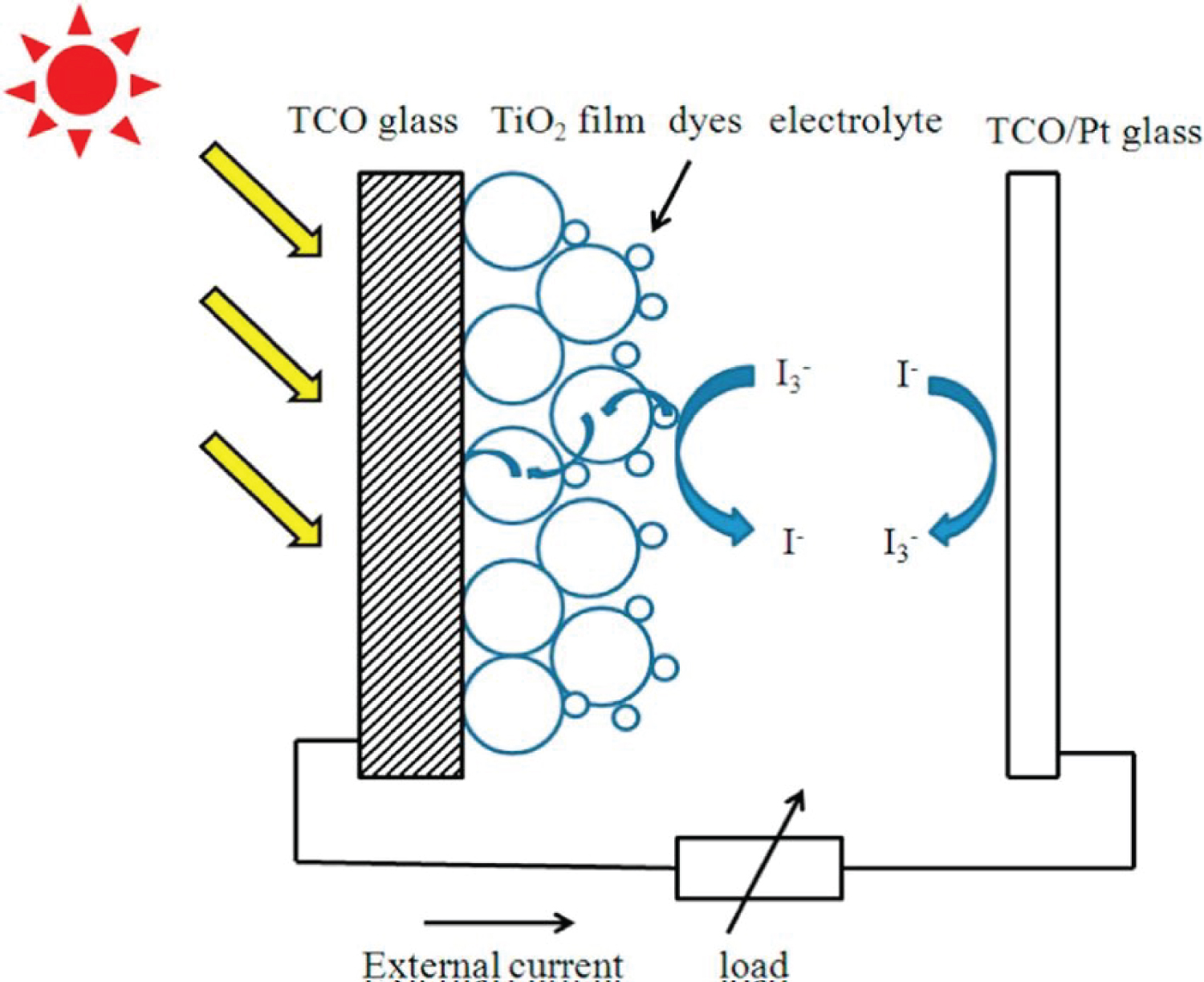
The dye sensitized solar cell consists of three main parts. At the top is a translucent glass to transmit light into it. Under the glass, TCO (Transparent Conductive Oxide), a conductive layer, is placed in order to collect the photoelectrons obtained in the anode (metal oxide and metal oxide-dye layers). Just below the TCO layer, there is a very thin nano-particle metal oxide layer (like TiO2). This layer has a fairly large surface area thanks to its nanostructure. Under the metal oxide layer, there is a dye layer with high photo activity. The dye layer is surrounded by a covalently bonded metal oxide layer for activity enhancement. Outside of this layer is the layer containing the electrolyte and redox couple. At the bottom of the cell, there is a permeable glass cathode (TCO) coated with platinum, which also contains a conductive layer. During the operation of the organic dye based photovoltaic cell, the net charge is always zero and no chemical change occurs. As a result of these electron transfer processes, photocurrent occurs. In DSSCs, continuous repetition of these reduction-oxidation processes is required to obtain continuous current. Performance measurements of DSSCs prepared using the dyes were carried out at Ege University Solar Energy Institute Laboratories. In the first stage, fluorine-added tin oxide (FTO) coated glasses were cleaned with detergent water, acetone and iso-propyl alcohol, respectively, and then 9-10 micrometer thick mesoporous anatase TiO2 was coated by screen printing method. TiO2/FTO electrodes were annealed at 550 ℃ for 45 minutes in order to obtain pure anatase TiO2 and to provide electronic connection between nano-crystals. The prepared electrodes were immersed in 0.5 mM solutions of the dyes prepared in THF and kept for 4 hours. In order to remove the dye molecules that are not chemically adsorbed on the TiO2 surface, it was first washed with its own solvent THF, then with DMF and acetonitrile respectively. For the counter electrode, FTO electrodes coated with a 1% solution of Hexachloroplatinic acid (H2PtCl6) were annealed at 470 ℃ for 30 minutes, and Pt4+ was reduced to Pt0 and Pt/FTO electrode was obtained. Pt/FTO electrode and TPDCX dye/TiO2/FTO electrode were glued together with the help of a 50 micrometer thick thermoplastic "surlyn 1702" frame in sandwich geometry. Finally, the ionic liquid derivative iodide/triiodide redox couple containing electrolyte was injected into the space between the two layers with the help of vacuum. The structure of the electrolyte contains 0.6M 1-butyl-3-methyl-imidazolium 51 iodide (BMII), 0.1M lithium iodide (LiI), 0.05 iodine (I2) dissolved in 3-methoxy propionitrile (MPN) and 0.5M tert-butyl pyridine (TBP). Photovoltaic characterizations were performed under dark and light (AM 1.5,100 mW/cm2) using a Keithley 2400 electrometer.
Results and Discussion
In solar cells with organic dye sensitizer, the position of the molecules relative to each other in the three dimensional plane and the interactions between molecules are of great importance for optimum charge transfer. According to the results here, it is seen that as a result of the attachment of different donor units to the molecule center, the molecule gains volume in three-dimensional plane relative to the center. According to the ground state (HOMO) and excited state (LUMO) charge densities of the dyes, it is understood that the charge density in the ground state is concentrated on the electron donor side groups. Therefore, the dyes are bound to the surface of the TiO2 through these groups. This situation can be considered as an important data that the dyes synthesized can show ideal behavior for DSSC applications.
Electrochemical Characterization of the Dyes
Bio Logic SP-50 model electrochemical study unit (potentiostat-galvanostat) was used in the electrochemical characterization of the dyes. In the cyclic voltammogram of the dyes, the peaks of the reducible in the structure were observed in the cathodic region. It belongs to the peak carboxyl group observed around -1.0 V in all voltammograms. The reduction belonging to the quinoxaline group was reversibly observed around Ered (1/2) = -1.5 V due to in-ring electron delocalization. As is known, oxidation occurs at the HOMO level of a molecule. A decrease in oxidation potential was observed with the addition of a second electron-donor group such as 3 hexylthiophene between molecule and donor groups. As a result, it was determined that the oxidation potential of the dyes increased with the increase in donor character and the HOMO-LUMO band gap value decreased from 1.95 eV to 1.77 eV (Figure 1, Figure 2 and Table 1).
Optical characterization of the dyes
Optical characterizations of the dyes were made using UV-Vis absorption spectroscopy. The π-π* transition of the molecule center group of dyes is in the range of 275-400 nm (Figure 3). The electron donor groups with different properties are attached to the molecule centered structure lead to create new extra charge transfer bands. In MC20 and MC28 dyes synthesized as a result of direct attachment of electro-donor groups to the molecule group, the charge transfer band was observed as same and the wavelength onset value does not shifted. The synthesized compounds are in the form of donor-acceptor and were used as photosensors in DSSC cells. The region with the highest solar spectrum is between 500-1200 nm. Therefore, organic dyes synthesized in recent years are desired to be absorbed in the near IR region.
As a result, Table 1 show that the Highest Occupied Molecular Orbital (HOMO), the Lowest Unoccupied Molecular Orbital (LUMO) of the dyes and their wavelength absorption. EHOMO = -(Eox-Eferrocene+4.8) eV and ELUMO= -(Ered-Eferrocene+4.8) eV. Here, 0.1 AgNO3 standard solution was used. Measurements were taken accordingly. Cyclic voltammetry of the dye in DMF (1 mM) was performed using a 5 mm diameter glassy carbon electrode (GCE), Ag/AgNO3 in acetonitrile and a platinum coil as the working, reference and counter electrodes, respectively. 0.1 M TBAPF6 was used as the supporting electrolyte. The Ag/AgNO3 reference electrode was calibrated using ferrocene (1 mM) in DMF. All the experiments were carried out in N2 saturated solutions using a CH660B potentiostat/galvanostat. Cyclic voltammetry experiments were used to determine the oxidation onset of each dye. Glassy carbon was used as the working electrode, Pt wire as the counter electrode, and Ag/AgNO3 as the reference electrode. Each sample was measured in 0.1 M TBAPF6 in anhydrous DMF and was calibrated with Fc/Fc+ as an internal standard. Oxidation onset values were then converted to NHE by the addition of 0.63 V.
Fabrication of DSSC device and their performance measurements
The results obtained from DSSCs prepared from the dyes that exhibit different electrochemical and optical behavior due to their different donor groups have shown us that the severe and wide absorption band resulting from the increase in donor character is highly effective on photovoltaic performance. Only 0.23% efficiency was obtained from MC20 dye, which has only the S atom differ in the molecule center and has a very low absorption band, while this efficiency was up to 0.25%, which contains S atom (Figure 3 and Table 1).
In the production of the solar cell, fluorine-doped conductive oxide coated transparent glass electrodes (Pilkington TEC-15; Surface: 15 Ω/sq) were used. Dye sensitized mesoporous structure TiO2 electrode working electrode; the platinum catalyst coated electrode was chosen as the counter electrode (Scheme 3). The surface of the working electrode was coated with TiO2 paste synthesized by sol-gel method using screen printing technique [31]. The coated electrode was sintered at 450 ℃ after drying at room temperature. The thickness of the TiO2 film was determined to be 10.3 µm on average (Tencor Alpha Step 500 profilometer). TiO2 coated electrodes MC-20 and MC-28 solution (DMF) prepared at a concentration of 5 × 10-4 M and standard Z907 cis-di isocyanato-(2 2'-bipyridyl-4,4'-dicarboxylic acid) - (2,2'-bipyridyl-4,4'-dinonyl) ruthenium (II) solution (acetonitrile: tert-butanol; 1: 1) by dipping the dyes overnight. It was adsorbed on semiconductor material. After the electrodes were removed from the dye solution, they were kept at 70 ℃ under vacuum and dried. The surface of the counter electrode was covered with a 1% hexachloroplatinic acid solution prepared in 2-propanol. After the electrode was dried, it was sintered at 400 ℃. The solar cell is in the shape of a sandwich, and "surlyn", a thermoplastic polymer with a thickness of 50 µm, was used to hold the electrodes together and provide the necessary space for the electrolyte. The electrolyte was filled into the cell under vacuum through the hole previously opened on the counter electrode. Electrolyte as 0.6 M N-methyl-N-butyl imidazolium iodide (BMII) + 0.1 M LiI + 0.05 M I2 + 0.5 M 4-t-butyl pyridine (TBP) prepared in 3-methoxypropionitrile has been prepared. The hole on the surface was closed using surlyn polymer. The active area of the produced solar cells was determined as 0.81 cm2.
Performance analysis of solar cells was performed using Keithley 2400 Source-Meter. The measurement was performed in AM1.5 standard (1.5 air mass and 100 mW/cm2 with 750 W Xe lamp) [31,32]. Keithley 2400 apparatus was used to obtain I-V curves of DSSC cells produced with the synthesized compounds. DSSC cells made Jsc, Voc, FF, Max. Parameters such as Power Point, Jmpp, Vmpp, and % Efficiency (Figure 4 and Figure 5) were calculated using a program written in Labview. The active area of the Solar cells was measured as 0.81 cm2.
The I-V characteristics of MC-20 and MC-28 show that the efficiency were measured as 0.23% and 0.25%, respectively (Table 2). Although the SNS group showed good donor characteristics, the efficiency of MC-20 and MC-28 remained very low even if the Jmpp (mA/cm2) was 300 and 320 for MC-20 and MC-28 and the Vmpp (mV) was 0.77 and 0.76, respectively (Table 2). This is thought to be due to the very low absorption in the visible region. On the other hand, the efficiency of cells made with both molecules is very close to each other. But the small difference can be explained as follows: Thiophene on MC-28 is both a good electron transfer and shifts its absorption to the red region.
Conclusion
In this study, a series of molecule-centered molecules with different HOMO-LUMO band gap values varying according to different donor and acceptor groups for DSSC applications were synthesized. The structures of all the synthesized electro active molecules have been elucidated by using 1H-NMR data. The optical band gap of the synthesized MC20 and MC28 electro active molecules was calculated with the onset values of UV-Vis absorption spectra. When the geometric optimizations of the dyes were examined, it was determined that the molecule group was planar in the center and the positions of the different donor groups connected to this group showed volumetricity in the three-dimensional plane. In addition, due to the donor acceptor arrangement of the structure, it is clearly seen that the charges are on the surrounding donor groups in the basic state (HOMO) and migrate onto the -COOH acceptor group on the molecule in the stimulated state (LUMO). Although the donor properties of SNSs are good, the absorption of MC derivatives in the visible region is very limited. Therefore, their efficiency remained very low. However, the first example molecules where the SNS functional group was used as electron donor in DSSC cells are MC-20 and MC-28. For this reason, it was evaluated that the synthesis and applications of new molecular structure designs that will shift the absorption spectrum on the molecule to the visible region and higher conversion efficiencies in photovoltaic cells may be possible. In addition, the electron transfer mechanisms of the sensitizing dyes synthesized within the scope of this work and applied on DSSC cells should be examined in order to clarify the photovoltaic performance problems they have shown. After performing the optical and electrochemical characterization of the synthesized the dyes, DSSC cells were prepared and the photovoltaic performance the change of the molecule central atom was investigated. As a result of the different atom at the molecule center does not change the photovoltaic efficiency too much. It is just increased from around 0.23% to around 0.25%.