
International Journal of Earth Science and Geophysics
(ISSN: 2631-5033)
Volume 5, Issue 2
Research Article
DOI: 10.35840/2631-5033/1830
Preliminary Studies on the Potential of Locally Available Shells in Sierra Leone for Phosphorous Removal
Ronnie AD Frazer-Williams*
Figures
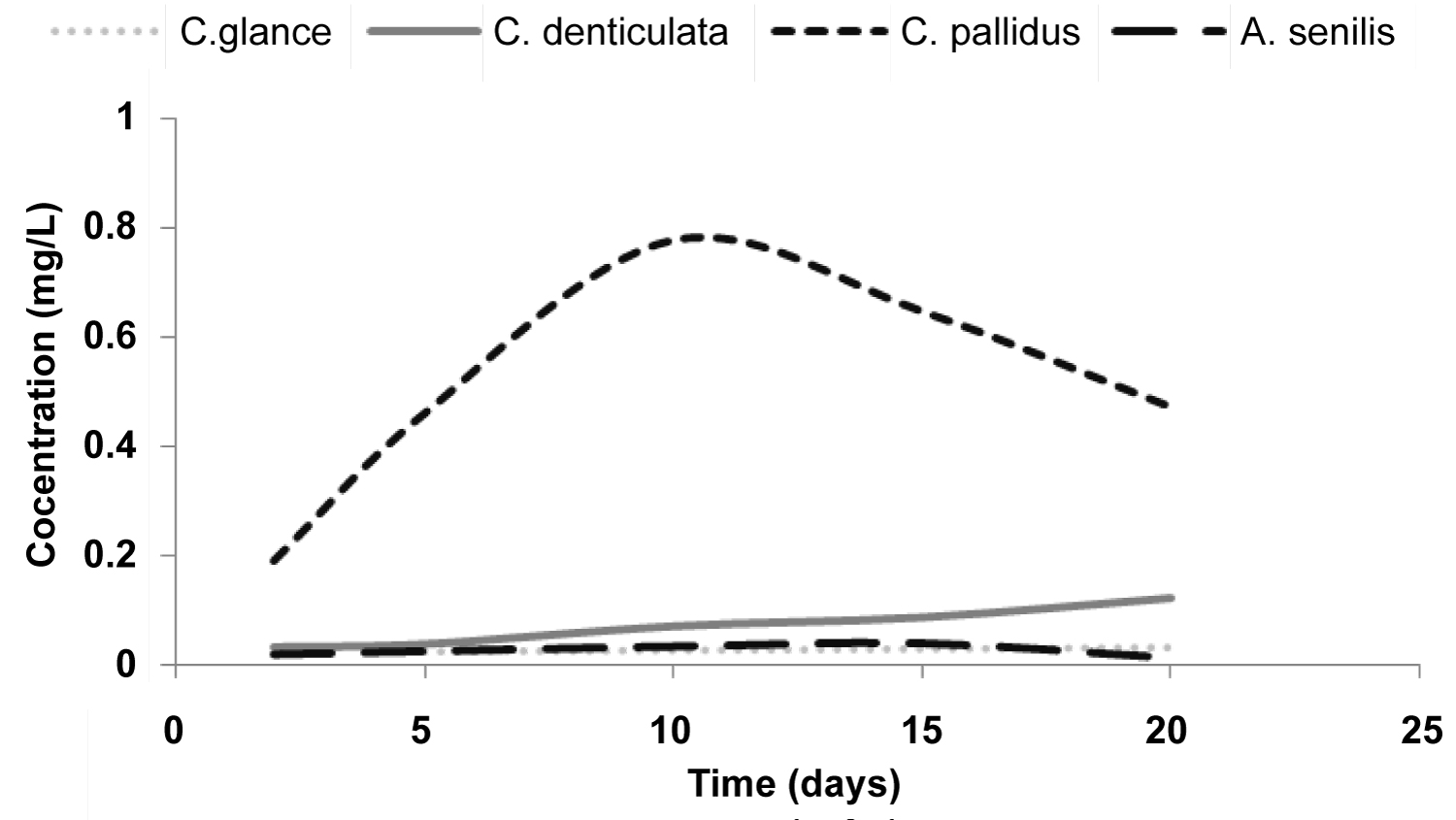
Figure 1: Graph showing the rate of release of Calcium....
Graph showing the rate of release of Calcium from various shells.

Figure 2: Pattern of uptake of phosphorous by....
Pattern of uptake of phosphorous by the various shells.
Tables
Table 1: Result of Ca leached by shells and pH of shell solution. Values represent average of two readings.
References
- Egbenda PO, Bangura A (1999) Soil structure modification by carbonate and oxide forms of Oyster shells. J Pure & Appli Sc 6: 39-45.
- Egbenda PO, Cole WS (1995) Investigation of the liming capacities of Oyster and another coral shell. Sierra Leone J Chem 10: 8-11.
- Del Bubba M, Arias CA, Brix H (2003) Phosphorus adsorption maximum of sands for use as media in subsurface flow constructed reed beds as measured by the Langmuir isotherm. Water Res 37: 3390-3400.
- Arias CA, Del Bubba M, Brix H (2001) Phosphorous removal by sands for use as media in subsurface flow constructed reed beds. Wat Res 35: 1159-1168.
- Molle P, Lie'nard A, Grasmick A, Iwema A (2003) Phosphorus retention in subsurface constructed wetlands: Investigations focused on calcareous materials and their chemical reactions. Water Sci Technol 48: 75-83.
- Xiong J, Qin Y, Islam E, Yue M, Wang W (2011) Phosphate removal from solution using powdered freshwater mussel shells. Desalination 276: 317-321.
- Westholm LJ (2006) Substrates for phosphorus removal-Potential benefits for on-site wastewater treatment? Water Research 40: 23-36.
- Von Wandruszka R (2006) Phosphorus retention in calcareous soils and the effect of organic matter on its mobility. Geochem Trans 7: 6.
- Søvik AK, Kløve B (2005) Phosphorous retention processes in shell sand filter systems treating municipal wastewater. Ecological Engineering 25: 168-182.
- Onoda H, Matsumoto R, Tafu M (2013) Removal of phosphorus with corbicula shells from resemble wastewater and adsorptive properties of by-products. IJEP 3: 1-4.
- Onoda H, Kawade H, Takenaka A (2012) Preparation of calcium phosphates from resemble phosphorus waste water and sea urchin shells. Journal of Ecotechnology Research 15: 107-111.
Author Details
Ronnie AD Frazer-Williams*
Department of Chemistry, Fourah Bay College, University of Sierra Leone, Sierra Leone
Corresponding author
Ronnie AD Frazer-Williams, Department of Chemistry, Fourah Bay College, University of Sierra Leone, Sierra Leone.
Accepted: December 28, 2019 | Published Online: December 30, 2019
Citation: Frazer-Williams RAD (2019) Preliminary Studies on the Potential of Locally Available Shells in Sierra Leone for Phosphorous Removal. Int J Earth Sci Geophys 5:030
Copyright: © 2019 Frazer-Williams RAD. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Four sets of locally available shells of Snail (Cymbium glance), Oyster (Crastrostrea denticulate), Crab (Callinectis pallidus) and Cockle (Senilia senilis) were studied for their potential to remove phosphorous from solution in a batch experiment. All four shells showed ability to release calcium in solution. The order of magnitude of calcium content contained in the four shells is in the order C. glance > S. senilis > C. denticulate >> C. pallidus. The resulting solution of the shells with the exception of S. senilis which was slightly acidic -neutral (6.58) was weakly alkaline (C. glance = 7.09, C. denticulate = 7.24 and C. pallidus = 7.29). Average P (g) per day removed by the shells were 0.016 for Snail (C. glance), 0.013 for Oyster (C. denticulate), 0.033 for Crab (C. pallidus) and 0.026 for Cockle (S. senilis) ranging from 13-33 mg P g-1. In addition to the total removal ability of the shells, the rate and extent of phosphorous removal by the shells followed the order: C. pallidus > S. senilis > C. glance > C. denticulate.
Keywords
Calcareous shells, Phosphorous removal
Introduction
A shell is the exoskeleton (hard protective outer layer) of an animal without a backbone (an invertebrate). Both freshwater and marine shells are basically composed of calcium carbonate. The level of calcium content per unit mass of calcium depends on the type and habitat of species.
Globally, the study of shells has been found interesting by the fact that shells have a wide range of application due to their different shapes, forms and constituents. For instance, based on their shape, they are used as decorations on the ground frontage of doors in the rural villages in Sierra Leone. Based on their strength, they have been used as tools e.g. some species of bivalves have been used as scrapers, blades scalps. Because shells are sometimes a ready available bulk source of calcium carbonate, shells such as Oyster shells are sometimes used as soil conditioners in horticulture as well as adsorbent in the removal of gases and desired ions from wastewaters as an environmental cleanup. To date, the use of calcareous shells in Sierra Leone has exclusively been studied in relation to agriculture. For instance, Egbenda and Bangura [1] demonstrated the usefulness of Oyster shells in bringing about structural modifications in soils with high amount of surface-active clays and low pH. Similarly, Egbenda and Cole [2], reported an improvement of the liming capacity of agricultural soils by Oyster and other coral shown found in Sierra Leone.
Shells have also been used as an adsorbent to absorb important and expensive industrial chemicals such as phosphorous in the form of phosphate from industrial and domestic wastewaters [3]. Although calcareous shells have been documented to be useful in removing P from wastewater [3], the nature of calcareous shells differ among and between locations. Hence, this could potentially influence their suitability for some applications such as substrate in a wastewater treatment plant.
The quest for finding suitable media for phosphorous (P) removal requires the selection of materials which must simultaneously have good hydraulic characteristics and sustained elimination of P from wastewaters. The initial determination of the inherent properties of the materials typically includes the characterization of the physical and chemical features. Once these parameters are considered, the selected material P capacity is then tested. Usually, sorption isotherm experiments which can indicate the P adsorption capacity of the media is conducted [4]. In view of the foregoing, this research primarily focuses on identifying shells with high calcium content and their ability to remove phosphorous from solution. Specific objectives of the study are: compare the level of calcium content of different shells (adsorbents) and how it relates to their phosphorous removal ability in solution and identify key physicochemical properties that will influence phosphate adsorption of the adsorbents.
Materials and Methods
Shells used for this study were identified and classified by their scientific names with the help of the Biological Sciences Department, Fourah Bay College, University of Sierra Leone. The four sets of shells used are Snail (Cymbium glance), Oyster (Crastrostrea denticulate), Crab ((Callinectes pallidus) and Cockle (Senilia senilis). Shells of Snail (Cymbium glance), Crab ((Callinectes pallidus) and Cockle (Senilia senilis) were collected from The Magazine Court Bay (a coastal bay along the eastern part of the capital city Freetown) whilst shells of Oyster (Crastrostrea denticulate) was collected from Calaba Town local White Wash factory situated at the outskirts of Freetown.
The shells were thoroughly washed with tap water to remove all dirt, sand and unwanted materials. Thereafter, shells were air dried at room temperature (~30 ℃) and stored in glass bottled until they were used for phosphorous uptake and Calcium (Ca) leaching studies. As the sizes of the shells were too big for batch studies in a beaker and conical flasks for such studies, shells used for phosphorous uptake were fragmented to sizes in the range 0.5 - 2.0 cm. Mixed sizes were subsequently used for batch studies. All determinations were carried out in duplicate.
All reagents used were of analytical grade (AR). Experiment to investigate the rate of release of Ca by the different shells was conducted. 5 g of each shell was weighed and placed in 100 ml beakers. 100 ml DI water was poured to cover the shells and left to stand for 15 days. 5 ml from each beaker containing the shells was withdrawn after 2, 5, 10 and 15 days. The solutions withdrawn were titrated with standard EDTA solution to determine the concentration of Ca leached from the shells in each of the solution over the 15 days interval.
Phosphorous stock solution was prepared by dissolving 4.39 g previously dried KH2PO4 salts in 100 ml distilled water. A 1.2% Ammonium molybdate reagent (m/v) was prepared by dissolving 3.75 g of Vitamin C tablets in 250 ml DI water. Phosphorous working standards were prepared from the stock solution as and when required. Phosphorous was determined spectrophotometrically using the Pekin Elmer Ultra Violet Spectrophotometer measured at 880 nm. Initial P concentration used for the batch studies was 0.44 g dm-3. pH of all solution was determined using the Thermo Orion SN: 057019, combine glass electrode pH meter after calibration of the instrument.
Results
The results of the amount of calcium leached from the various shells when immersed in tap water for 24 hours is presented (Table 1). Shells of C. glance, C. denticulate and S. senilis are generally comparable but much higher than shells of C. pallidus.
Solutions of C. denticulata and C. pallidus gave alkaline pH and are also slightly higher than those of C. glance and S. senilis. The pH of S. senilis can be described as very weakly acidic or almost neutral.
Result of the rate of release of calcium from shell is also presented (Figure 1). Overall, rate and amount of release of calcium was higher for C. pallidus than the other three species. Rate of Ca released from C. pallidus was high and fast but came to a halt with an eventual decline after day 10 (Figure 1). The other three species showed a gradual and continual but much lower rate of release of calcium.
Figure 2 show the pattern of uptake of phosphorous by the various shells. In general, the uptake of phosphorous by the shells is in the order Crab (C. pallidus) > Cockle (S. senilis) > Snail (C. glance) > Oyster (C. denticulate). Average P (g) per day removed by the shells were 0.016 for Snail (C. glance), 0.013 for Oyster (C. denticulate), 0.033 for Crab (C. pallidus) and 0.026 for Cockle (S. senilis), i.e. ranging from 13-33 mg P g-1. The shells of snail, crab and cockle almost gave a linear plot between shell mass of 2.5 to 10g except for Oyster (C. denticulate) which shows a continual increase. Between masses 12.5 and 15 g, shells of Crab and Cockle increase dramatically.
Discussion
All solutions experienced an increase in pH, albeit a small increase. Despite the increase in pH, all values are close to neutrality and well within discharge limits of 6-9 into natural waters. Possible reasons for the increase in pH could be as a result of the release of carbonate ions from the shell (equation 1) and the eventual formation of hydroxide and bicarbonate in a subsequent reaction of carbonate ions in water (equation 2). Alkaline pH of treated wastewater from calcareous substrate is typical. However, such alkaline nature may cause an adverse effect to the receiving aquatic ecosystem [5].
The order of magnitude of calcium content contained in the four shells was in the order C. glance > S. senilis > C. denticulate >> C. pallidus. C. pallidus having the least amount of leachable calcium, showed the highest rate of release of calcium in solution. The fact that C. pallidus contains the least amount of calcium content compared to the other three shells (Table 1) possibly explains why the available Calcium in solution from C. pallidus reaches a maximum within a half of the time the other three shells take to reach their maximum. Result of removal capacity in this study ranging from 13 - 33 mg P g-1 exceeded some reported literature values. For instance, in a study using freshwater mussel shells in a laboratory-scale batch experiments, Xiong, et al. [6] reported effective P removal saturation adsorption capacity of 6.95 mg P g-1. However, comapring data between researches should be done with caution since there are no researchers who have caried out investigations exactly the same way [7].
Although detailed mechanistic studies were not investigated in this work, studies on the interaction of phosphorous solution with calcareous surfaces is well documented. Results from the literature revealed removal of phosphorus by surface reaction/adsorption and co-precipitation mechanism [3,8,9]. The role of calcium carbonate in P retention/removal by calcareous soils is significant only at relatively high P concentrations with non-carbonate clays playing a more important part at lower concentrations. This study was conducted under high P concentration with an adsorbent virtually composed of calcium carbonate. Hence, it stands to reason therefore that this study supports the important notion that calcium carbonate plays a dominant role of P removal from solution at alkaline condition. The mechanistic reason for this is because at alkaline condition, the formation of calciumphosphate precipitate is kinetically and energetically favorable. In two separate studies using Corbicula shells [10] and Oyster shells [11] (both composed mainly of calcium carbonate), X-ray diffraction (XRD) technique confirmed the formation of calcium hydrogen phosphate di-hydrate (CaHPO4.2H2O) from a reaction between the shells and phosphoric acid. In addition, P sorption on calcareous surfaces depends on surface characteristics, especially surface area and zeta potential (17.7 - 25.3 mV) [8].
Results of this study further indicates that the removal of phosphorous from all shells generally does not increase significantly by continuously increasing adsorbent quantity (Figure 2). This is important in that in wastewater treatment plant applications, shell-based plants could be designed that utilizes smaller space in order to achieve maximum results, especially for countries where available space is limited.
Conclusion
The study generally showed that locally available shells have the potential of removing phosphorous from solution. Such positive results is a first step in investigating materials as suitable adsorbent in wastewater treatment as other parameters such as the adsorbent hydraulic conductivity, porosity, pH of the solution in the treatment plant, strength of the adsorbent influence the suitability of an adsorbent in removing phosphorous from constructed wetland wastewater treatment plant. Further work on different grain sizes at varying pH conditions in the presence of competing ions such as organic ligands will form the next phase of this study using wastewater instead of tap water dosed with phosphate.

